Stomach cancer atlas maps path to better treatments
Scientists in Singapore have assembled the world's largest and highest-resolution atlas of gastric cancer, a leading cause of global cancer death. The atlas provides, at the level of single cells, new insights into how gastric cancers progress and how tumors from separate patients differ from one another. The findings also identified molecular pathways driving gastric cancer growth and spread to other organs, and also how the disease can be better treated.
The research—led by Duke-NUS Medical School and the Agency for Science, Technology and Research (A*STAR)'s Genome Institute of Singapore (GIS), and involving multi-institutional collaborators, was published in Cancer Discovery, providing an essential reference for experts tackling one of the most common cancers in Asia.
"Patients with gastric cancer—also referred to as stomach cancer—are notoriously difficult to treat because we lack a clear understanding of the types of cells that make up their tumors," said the study's principal investigator, Professor Patrick Tan, from Duke-NUS' Cancer & Stem Cell Biology Programme and GIS. "With precision oncology, cutting-edge technologies like single-cell sequencing can analyze patient cancers at unprecedented depth and resolution, to tailor therapies for patients and reveal new pathways to improve diagnoses and treatments." Prof Tan is also a Senior Principal Investigator at CSI Singapore.
"This study is important because it provides new insights into how gastric cancer develops. It is the most comprehensive study of its kind using advanced research technologies. Excitingly, our study identified some novel drug targets that may pave the way for new treatment strategies," said Professor Jimmy So, Head and Senior Consultant at the National University Hospital (NUH)'s Division of General Surgery (Upper Gastrointestinal Surgery). He is also Professor of Surgery at NUS Yong Loo Lin School of Medicine (NUS Medicine), and the Head and Senior Consultant at the Division of Surgical Oncology, National University Cancer Institute, Singapore.
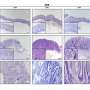
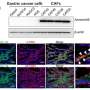
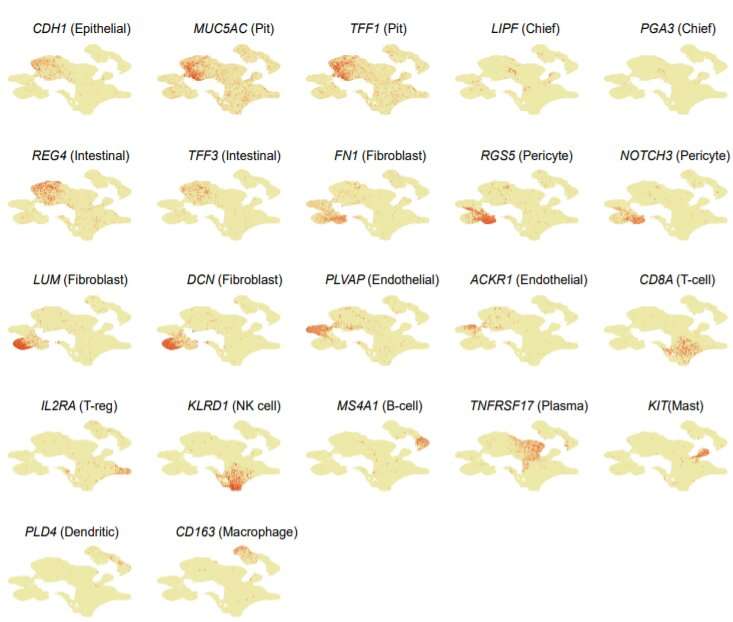
This work was performed as part of the Singapore Gastric Cancer Consortium (SGCC), a national body of clinicians and scientists working in translational research on gastric cancer supported by the National Medical Research Council. The research team sampled 46 tumors from 31 patients at various stages of the disease. From these samples, they analyzed 200,000 separate cells, using innovative techniques to assess individual patterns of gene expression.
"This powerful new cell atlas, enabled via team-based science, illuminates the path to novel therapies and further research in gastric cancer," said Professor Yeoh Khay Guan, Lead Principal Investigator of SGCC, and the Chief Executive of NUHS. He is also a Senior Consultant with the NUH Division of Gastroenterology and Hepatology, and the Irene Tan Liang Kheng Professor in Medicine and Oncology, NUS Medicine.
The scientists found more than 30 different cell types, some of which had not been detected in gastric tumors before. Tumors from different patients showed a different composition of these cells, which helps to explain why similar treatments can have very different outcomes.
Some types of gastric tumor were found to contain immune cells, particularly B cells that control the generation of antibodies. This suggests that cancer immunotherapy, a type of treatment that uses a person's own immune system to fight cancer, could be applied to the disease. The study also showed a previously unknown role for common cells called fibroblasts in driving gastric cancer. Targeting these cells could offer new avenues for treatment.
Dr. Shyam Prabhakar, associate director, spatial and single cell systems, GIS, said, "This work puts the power of single-cell sequencing to very effective use by creating the first in-depth map of cell types in stomach cancer. Now that we have the 'parts list," we can identify the vulnerabilities of these tumors and start to target them."
Professor Patrick Casey, senior vice-dean for research at Duke-NUS, said, "While gastric cancer is common in Asia, it is relatively rare in the West. This has led to scientific disparities where little is known about the driving pathways, compared with diseases like breast and colorectal cancer. This work therefore underscores the critical importance of supporting scientists doing research on Asian-specific conditions."
Building on this work, the research team is currently investigating how the cell populations identified in the study change over time, either during early cancer development or as a consequence of drug treatment. This information may yield pathways that can be exploited for early cancer detection and drug development.
Furthermore, because many of the features identified in this study may be applicable to other cancer types. The team will collaborate with researchers working on other cancers to pool data and identify cell populations that contribute to multiple cancers.
Gastric cancer remains a significant cause of cancer death in Singapore, particularly in males, among whom it is the fourth highest cause of cancer death. Veteran radio DJ and pop culture personality Chris Ho died of the disease earlier this year.
More information: Vikrant Kumar et al, Single-cell atlas of lineage states, tumor microenvironment and subtype-specific expression programs in gastric cancer, Cancer Discovery (2021). DOI: 10.1158/2159-8290.cd-21-0683