New study reveals how epithelial cells in the body naturally eliminate 'precancerous' ones
In addition to its immune surveillance system, recent reports have shown that the human body has defense mechanisms run by non-immune epithelial cells. Epithelial cells are a type of cell that occur in layers that line most surfaces of our body. These epithelial cells can recognize and extrude neighboring precancerous cells from the epithelium; this extrusion process is called cell competition. This form of immune-like surveillance has garnered attention in recent years based on its potential for future immune-like therapeutic targets for cancer preventive treatment. However, it is still unknown what kind of ligand-receptor interactions are involved in the recognition of precancerous cells by normal epithelial cells. Recently, a group of scientists have successfully solved this research question and have published their findings in Nature Immunology.
Speaking about the premise of their study, Professor Takeshi Maruyama, an Associate Professor at the Waseda Institute for Advanced Study at Waseda University, who led the research group, says that "during the process of cell competition, normal epithelial cells can be primed by contact with precancerous cells. However, it was previously unclear how neighboring normal epithelial cells recognize precancerous cells to eliminate them."
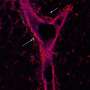
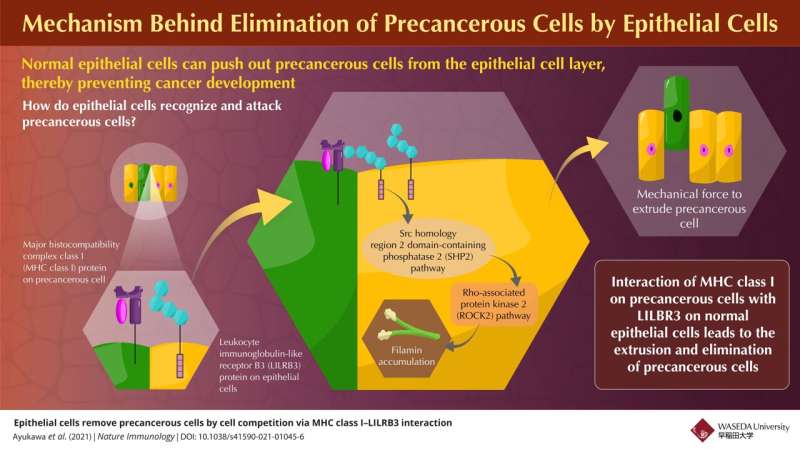
In this work, the researchers identified a plasma membrane protein, Canis suboptimal alteration recognizing protein (Canis AltR) in non-transformed canine epithelial cells, the function of which was unknown, as a recognizing protein for cell competition. In humans, the protein most similar to AltR is leukocyte immunoglobulin-like receptor B3 (LILRB3). AltR/LILRB3 interacts with major histocompatibility complex class I (MHC class I) that is expressed on precancerous epithelial cells.
MHC class I-AltR/LILRB3 interaction causes the activation of AltR/LILRB3, which triggers an intracellular SHP2–ROCK2 (Src homology-2 domain-containing protein tyrosine phosphatase-2–Rho-associated protein kinase 2) pathway. This SHP2–ROCK2 pathway leads to the "accumulation of cytoskeletal components," which generates a mechanical force to extrude precancerous cells, in the normal epithelial cells at the boundary with precancerous cells. Finally, normal epithelial cells push the precancerous cells out of the epithelium to eliminate them from the body.
Interestingly, this molecular mechanism occurs independently of natural killer or CD8+ T cell-mediated immune responses. "Our study describes a new immune-like mechanism by non-immune epithelial cells to suppress tumorigenesis," says Maruyama.
The researchers hope that these significant findings can be applied to real life cancer treatment. Maruyama adds that "the recombinant MHC-I-α3 protein used in this study enhances the elimination of precancerous cells and suppresses the formation of tumors and precancerous lesions. We hope that this biomolecule would contribute to a therapeutic candidate for cancer prevention by the elimination of precancerous cells."
More information: Shiyu Ayukawa et al, Epithelial cells remove precancerous cells by cell competition via MHC class I–LILRB3 interaction, Nature Immunology (2021). DOI: 10.1038/s41590-021-01045-6