Immunotherapy drug shows promise in advanced endometrial cancer
A cancer immunotherapy drug currently approved by the U.S. Food and Drug Administration (FDA) to treat several forms of cancer is also effective for treating aggressive forms of endometrial (uterine) cancer, according to results from an international phase II clinical trial led by researchers at The Ohio State University Comprehensive Cancer Center-Arthur G. James Cancer Hospital and Richard J. Solove Research Institute (OSUCCC-James).
For this study, researchers enrolled 90 women diagnosed with recurrent or advanced endometrial cancer to determine whether the drug pembrolizumab (marketed as Keytruda) could be used to effectively treat this subset of patients with MMR deficient (dMMR) or MSI-high tumors. The study included patients treated at 38 hospitals in 15 countries.
Pembrolizumab is a cancer immunotherapy that works by inhibiting certain cellular receptors that prevents the immune system from recognizing and destroying cancer cells. The FDA has approved the drug for treating several other cancers, including melanoma, lung, head/neck, cervical and stomach cancer.
In this study, researchers showed that 48% of advanced endometrial cancer patients experienced a complete or partial response. Two thirds of these patients also had a response that lasted more than three years. Additionally, two thirds of all patients in the study had a clinical response. This trial is the longest follow-up reported to date, with a median follow up of 42.6 months.
"These findings suggest a long-term benefit to patients. Even the potential for curative intent is now possible in patients with recurrent or metastatic uterine cancer," said Dr. David O'Malley, lead author of the study and a gynecologic oncologist at the OSUCCC-James.
Clinical trials to evaluate pembrolizumab for the treatment of earlier stage disease are currently underway.
The international research team reports its findings online first Jan. 6, 2022, in the Journal of Clinical Oncology.
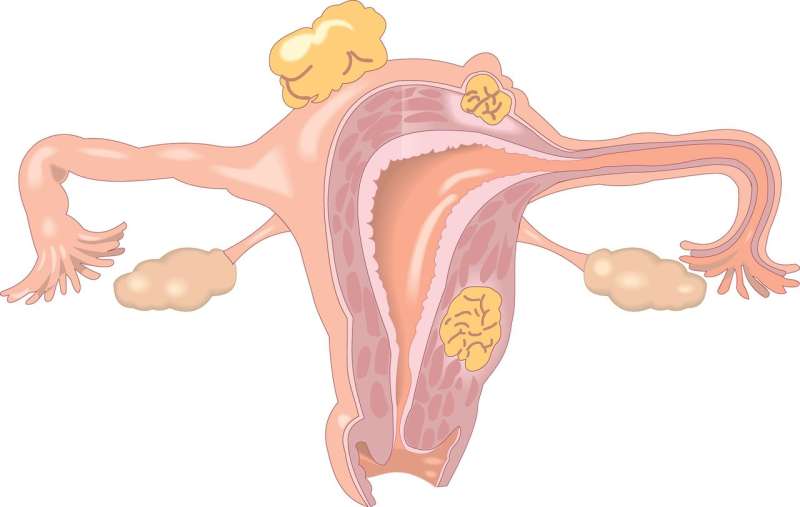
Endometrial cancer is the second most common form of cancer in women worldwide, and incidence rates have been increasing. While platinum-based chemotherapy regimens can be effective, many patients experience a recurrence. Effective second-line therapies for recurrent or metastatic disease are limited, resulting in a 17% five-year survival rate for women with advanced or recurrent disease.
Previous research suggests that up to 31% of patients with endometrial cancer have DNA structure changes known as microsatellite instability (MSI-H) and mismatch repair deficiency (dMMR). In a group of patients (approximately 2%) these are inherited mutations (passed down through families, clinically known as Lynch syndrome) in one of the mismatch repair (MMR) genes. Dr. O'Malley notes that the mutation is usually present in the tumor and not a genetic mutation.
"By targeting this damaged pathway with this targeted drug, we can reset the cellular mechanisms and allow the immune system to reactivate and attack the cancer cells," said Dr. O'Malley.
Prior to the data from this study, there was no accepted standard-of-care second-line therapy for endometrial cancer patients with MSI-H/dMMR-positive tumors; however, researchers note that the overall response rate for this study is very strong when compared to the 10-15% expected response rate resulting from other chemotherapy agents that are most often used as second-line therapy.
More information: David M. O'Malley et al, Pembrolizumab in Patients With Microsatellite Instability–High Advanced Endometrial Cancer: Results From the KEYNOTE-158 Study, Journal of Clinical Oncology (2022). DOI: 10.1200/JCO.21.01874