Feeding mode of newborns could influence oral bacteria makeup
After birth, the human mouth quickly becomes a hotbed of microbial variation. Streptococcus species largely dominate the oral cavity for the first six weeks of life, but the bacterial population diversifies with age and experience. Researchers study this early development, in part, to understand connections between the oral microbiota and associated diseases.
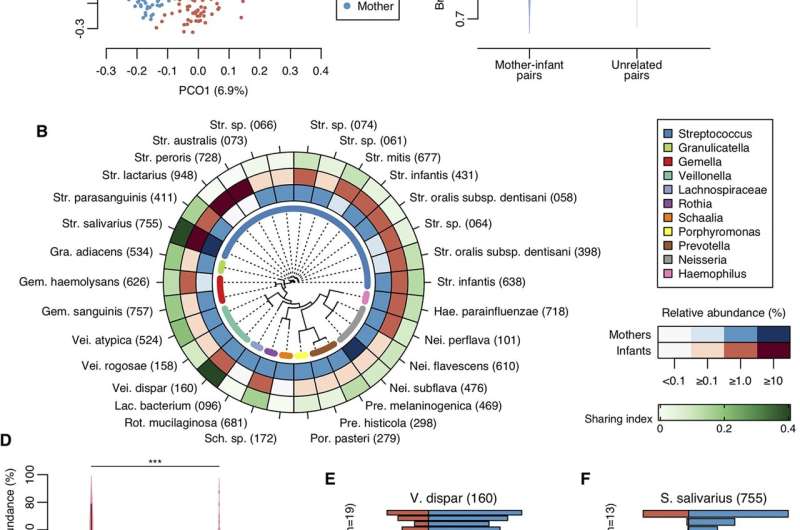
An infant's mother is likely a major source—if not the most important one—of the early oral microbiota. This week in mBio, dental researchers in Japan report a new analysis of how new mothers share microbes with newborns. The researchers collected 892 tongue samples from 448 pairs of moms and babies (217 males, 231 females), collected when the children were 4 months old, to measure bacterial abundance and, more specifically, the abundance of single, unique DNA sequences, called amplicon sequence variants (ASV), shared between mother and child.
Shared ASVs in newborns ranged from almost none to almost 100%, said Yoshihisa Yamashita, Ph.D., at Kyushu University in Japan, senior author of the study. "The acquisition level of maternal oral bacteria varied widely among individuals," he said. The median relative abundance of ASV that newborns shared with their mothers, however, was 9.7%, which the researchers noted was significantly higher than the abundance of ASVs newborns shared with other, unrelated mothers. The study was led by Shinya Kageyama, Ph.D, also at Kyushu University.
Notably, the shared abundance and composition varied significantly by how the infant fed. Infants who breastfed exclusively shared fewer ASVs with their mothers than infants fed exclusively with formula or fed with some combination of breastfeeding and formula. Breastfed infants also had a bacterial composition most unrelated to the mothers compared to other groups. The researchers found no difference in abundance linked to the age, sex, delivery mode, family smoking status or antibiotic use of the infants.
The researchers offered two hypotheses to explain the difference. "One is that protective factors of breast milk regulate mother-derived oral bacterial colonization," said Yamashita. The second is that the different substrates provided by formula and breast milk influence the balance of bacteria in the mouth.
Unlike previous studies that have reported shared microbes between mothers and infants, the new work includes a full-length analysis of all nine hypervariable regions in the 16s rRNA gene. In addition, the authors noted that the ASV approach allows for the identification of DNA sequences that differ by as little as a single nucleotide.
At Kyushu University, the researchers focus on connecting early-life oral microbiota to the risk of disease later in life. Previous studies have linked certain bacteria to cavities in the teeth and periodontitis. More recent research, however, has also reported that microbes usually found in the mouth show up in the gut in people with serious conditions ranging from liver cirrhosis to inflammatory bowel disease to colorectal cancer.
The new findings, said Yamashita, are "vital as a foundation for further studies." The group plans to follow the same study population, with the next examination of the oral microbiota and clinical conditions set for the infants' 3-year checkups.
More information: Shinya Kageyama et al, High-Level Acquisition of Maternal Oral Bacteria in Formula-Fed Infant Oral Microbiota, mBio (2022). DOI: 10.1128/mbio.03452-21