Spleen function discovery could lead to better treatments for infectious diseases
Researchers at the Peter Doherty Institute for Infection and Immunity (Doherty Institute) have discovered a new gene that plays an important role in the way the spleen functions, potentially leading to new treatments for infectious diseases.
The study, published in Science Immunology, also uncovered multiple new spleen cells and revealed the distinct way they respond in order to fight off different infections.
The spleen plays a key role in the immune responses that protect the body against various diseases and infections such as virus infections, malaria and sepsis, and also plays a key role in the immune response to vaccines. However, it has not been known how the spleen functions to support this response.
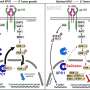
University of Melbourne Professor Scott Mueller, a Laboratory Head at the Doherty Institute and lead author on the paper, explained that while it is known the spleen is made up of various networks of cells called fibroblasts, a clear picture of how these cells are constructed and function, was lacking.
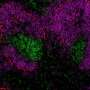
"Using novel biological tools and next generation sequencing, we were able to examine precisely how specialized types of fibroblast cells dictate how the spleen works to protect against infections," Professor Mueller explained.
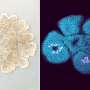
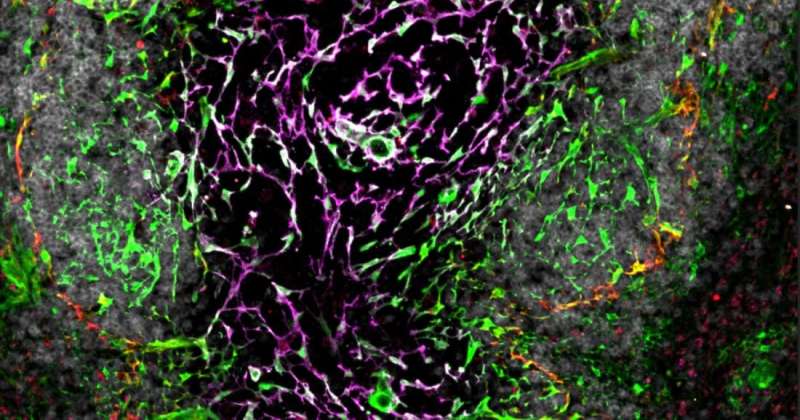
"We performed next-generation sequencing to understand what genes are expressed by fibroblasts in the spleen. We used advanced fluorescent microscopy to visualize the 3D networks of cells in the spleen.
"Since the fibroblasts are the cells that construct the architecture of the spleen, or the building blocks that make the tissue, the combination of advanced approaches allowed us to discover how these cells function and support immune responses to infections. One major outcome of this was the discovery of the gene SpiB, which plays a previously unknown role in spleen development and immunity."
Professor Mueller said this work provided a new roadmap of the spleen that could lead to new therapies to treat diseases such as severe acute virus infections and chronic infections, and new ways to target the immune system to improve disease outcome.
"By surveying the fibroblast landscape of the spleen and providing the first in-depth look at the gene expression, we have been able to create detailed plans to the scaffold of this unique organ," Professor Mueller said.
"These plans can now be utilized to discover new targets to treat diseases involving the spleen, as well as enable the design of more efficient vaccines."
More information: Yannick O. Alexandre et al, A diverse fibroblastic stromal cell landscape in the spleen directs tissue homeostasis and immunity, Science Immunology (2022). DOI: 10.1126/sciimmunol.abj0641