Molecular networks could explain racial disparity in triple negative breast cancer deaths
Different activity in two molecular networks could help explain why triple negative breast cancers tend to be more aggressive in African American (AA) women compared with white American (WA) women, a new study led by Johns Hopkins Kimmel Cancer Center researchers suggests.
The findings, published online Dec. 20 in eLife, may explain the marked racial disparity in mortality for this subset of breast cancer and offer new strategies for treating this deadly disease.
"The incidence of breast cancer in African American women is lower than in white American women, but mortality is higher. Our work shows the molecular mechanisms that cause cancer cells to grow and spread faster in African American women," says study leader Dipali Sharma, Ph.D., professor of oncology at the Johns Hopkins University School of Medicine and John Fetting Fund for Breast Cancer Prevention researcher.
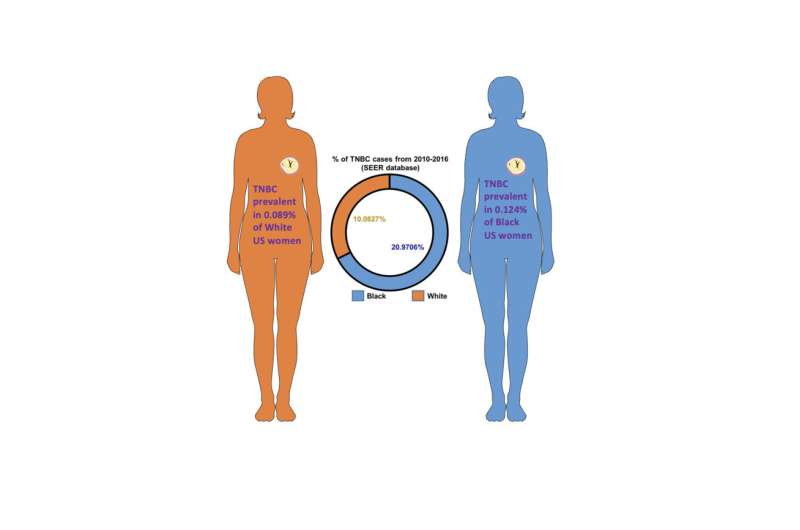
Better diagnosis and treatment have decreased breast cancer mortality in the past few decades, cutting death rates by about 40%, but AA women are still 42% more likely to die from this disease than WA women. Although AA women have a lower incidence of breast cancer than WA women overall, the disparity in mortality can be partially explained by a higher incidence among AA women of triple negative breast cancer, an aggressive breast cancer subset characterized by a lack of estrogen, progesterone and HER2 receptors. However, even among triple negative breast cancers, those in AA women tend to be even more aggressive than those in WA women, significantly decreasing AA survival rates.
To better understand this phenomenon, Sharma and her colleagues compared the behavior of triple negative breast cancer cell lines isolated from AA and WA women. When grown in petri dishes, the cells from AA women multiplied at a faster pace, were more invasive to surrounding tissue, were more likely to migrate, and had more "stem-like" cells capable of creating new tumors compared with the cells from WA women. When placed into mice, the cells from AA women created larger tumors faster and metastasized quicker compared with the cells from WA women.
"The triple negative breast cancer cells from African American women were more aggressive from the get-go," says Sumit Siddharth, first author on the paper and postdoctoral fellow at Johns Hopkins Medicine.
Searching for the molecular mechanisms behind these differences, the researchers assessed gene activity in each of the cell lines and in tumor samples from AA and WA women. They found that two genes, known as GLI1 and Notch1, were more active in cells isolated from AA women compared with those isolated from WA women. Both genes produce proteins known as transcription factors, explains Sheetal Parida, a study co-author and postdoctoral fellow, and are responsible for regulating large networks of other genes that can broadly affect cell function.
To determine whether GLI1 and Notch1 could serve as effective targets for triple negative breast cancers, the researchers dosed the cell lines from WA and AA women with an inhibitor for each of these two genes along with a common chemotherapy agent used to treat these cancers. Although each agent was largely ineffective on its own, combining all three drugs significantly inhibited growth, invasion and migration in the AA cell lines. Treating mice with tumors grown from these cell lines had similar positive effects, reducing tumor size and metastasis.
Sharma says that she and her team plan to continue to test GLI1 and Notch1 inhibitors in mouse models of triple negative breast cancers to find the best combination with chemotherapies and eventually investigate this strategy in clinical trials of patients with this disease subtype.
"Eventually," she says, "we may be able to close the survival gap between African American and white American women with triple negative breast cancers."
More information: Sumit Siddharth et al, Concomitant activation of GLI1 and Notch1 contributes to racial disparity of human triple negative breast cancer progression, eLife (2021). DOI: 10.7554/eLife.70729