AI drives quest for new antivirals to fight outbreaks
Research into drugs to treat mosquito-borne flaviviruses such as Zika and dengue as well as COVID-19 will benefit from a major funding boost, says a group of international scientists using artificial intelligence to discover new oral antivirals.
A research consortium led by the non-profit COVID Moonshot has been awarded more than US$68 million from the U.S. National Institutes of Health (NIH) to discover and develop globally accessible and affordable novel oral antivirals to combat COVID-19 and future pandemics.
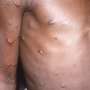
The development comes as monkeypox outbreaks have been declared around the world, raising concerns about the rapid spread of such viruses. Monkeypox is a viral disease that the World Health Organization says has emerged in at least 23 countries where the disease is not regularly found since 13 May.
The open-science COVID Moonshot was established in 2020 with the objective of developing a safe, globally accessible and affordable antiviral pill for COVID-19.
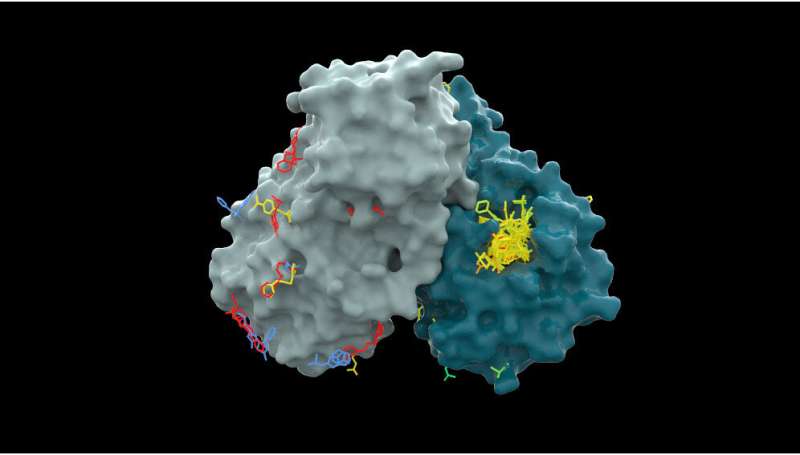
The consortium has created the Artificial Intelligence-Driven Structure-Enabled Antiviral Platform (ASAP) to build an antiviral discovery pipeline using structural biology, machine learning and computational chemistry.
The Drugs for Neglected Diseases initiative (DNDi), a founder of COVID Moonshot, says that the initial three-year phase of the ASAP project will aim to produce preclinical candidates against viral families that have been historically neglected by the market, with an initial focus on coronaviruses.
ASAP will also address flaviviruses, responsible for large endemic diseases such as dengue and Zika, DNDi says.
Rachel Cohen, executive director of DNDi North America, told SciDev.Net that the consortium was committed to ensuring "equitable and affordable access to any antivirals that may be developed as a result, especially in Africa, Asia, Latin America."
Cohen said that if preclinical antiviral candidates had been ready for clinical trials in humans when COVID-19 struck in late 2019, millions of lives might have been saved. She believes equitable access to antivirals in low- and middle-income countries should be a global priority.
"In our view, there can be no geographic restrictions in who would be able to access novel antivirals, and they must be affordable for both the individuals who need them and the health systems that serve them," said Cohen.
Francisca Mutapi, co-director of the Global Health Academy at the University of Edinburgh in Scotland, says that any development that supports the production of accessible drugs for infectious diseases is welcome for low- and middle-income countries, which carry the highest global burden of infectious diseases.
But Mutapi, who is not involved in the project, told SciDev.Net the consortium should include members from low- and middle-income countries, particularly Africa.
"Africa needs to be growing its capacity to discover and produce drugs and vaccines not only for Africa, but to contribute solutions for global health security," said Mutapi.
Babatunde Salako, director general of the Nigerian Institute of Medical Research, said he hoped the AI-driven platform would help increase the weaponry available to the world's medical workforce to fight emerging diseases. But he said capacity would need to be increased in the global South before lower-income countries could take advantage of the project's developments.