Hope for Alzheimer's blood test
Researchers from Macquarie University's Centre for Ageing, Cognition and Wellbeing are one step closer to a blood test to diagnose Alzheimer's early in the disease's progression.
Professor Ralph Martins and colleague Dr. Pratishtha Chatterjee are lead authors on a paper in Alzheimer's & Dementia showing that several blood biomarkers reflect the core hallmarks of Alzheimer's disease, which is the most common form of dementia.
Professor Martins says current standard clinical testing only provides a possible or probable diagnosis of Alzheimer's.
"Cerebrospinal fluid samples and brain imaging that can confirm if someone has Alzheimer's, but these are invasive and expensive, so they are not commonly done," he says.
"Blood biomarkers would be cheap, easily accessible and have the ability to deliver high throughput testing."
"It's important to be able to confirm a diagnosis early on, as this will allow patients and their families to be better prepared for future challenges, provide opportunities for them to be involved in clinical trials, and lower the cost of screening participants for these trials."
The study makes use of data from the Australian Imaging Biomarker and Lifestyle Study of Aging (AIBL).
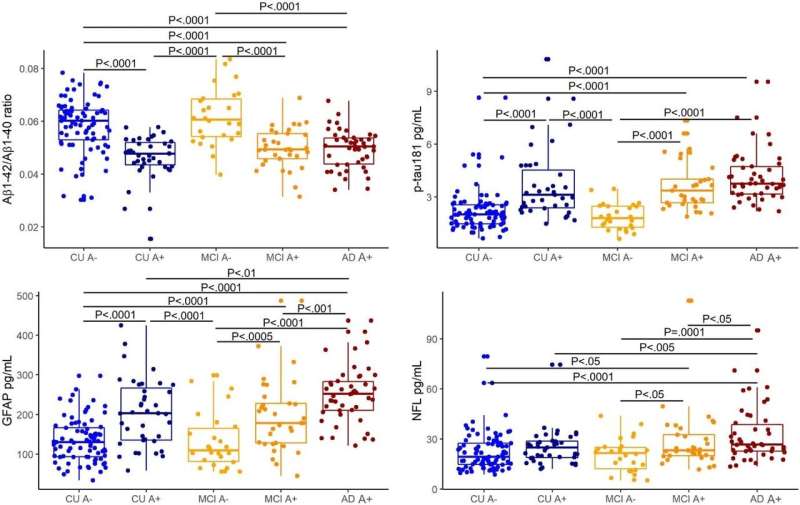
Dr. Chatterjee says they found that a panel of blood-based biomarkers, including amyloid beta (Aβ42/40 ratio), phosphorylated-tau181 (p-tau181), and glial fibrillary acidic protein (GFAP), had high discriminative performance for Alzheimer's disease from preclinical to dementia stages with an accuracy of 85 to 95 percent.
"We also showed that low plasma Aβ and high p-tau181, GFAP and neurofilament light chain (NFL) levels were associated with faster future cognitive decline, and low plasma Aβ and high p-tau181 and GFAP were associated with faster future brain Aβ accumulation," she says.
"Over 36 months, plasma Aβ decreased, and p-tau181 and GFAP increased, at a faster rate in people with mild cognitive impairment when compared to healthy people, and GFAP and NFL increased at a faster rate in Alzheimer's disease when compared to healthy individuals."
Dr. Chatterjee says further investigation will now be required to validate the clinical cut-off points for implementation in clinical settings, including looking at people from multiple ethnic backgrounds and those with comorbidities.
More information: Pratishtha Chatterjee et al, Plasma Aβ42/40 ratio, p‐tau181, GFAP, and NfL across the Alzheimer's disease continuum: A cross‐sectional and longitudinal study in the AIBL cohort, Alzheimer's & Dementia (2022). DOI: 10.1002/alz.12724