Scientists zero in on genetic causes of Parkinson's

Variants of at least 20 different genes have been closely linked to the development of Parkinson's disease, but scientists are still investigating how exactly they cause the severe and incurable motor disorder that afflicts about 1 million people in the U.S. alone.
New research by Yale researchers offers important clues. In two new papers, scientists provide insight into the function of a protein called VPS13C, one of the molecular suspects underlying Parkinson's, a disease marked by uncontrollable movements including tremors, stiffness, and loss of balance.
"There are many roads to Rome; likewise there are many roads leading to Parkinson's," said Pietro De Camilli, the John Klingenstein Professor of Neuroscience and professor of cell biology at Yale and investigator for the Howard Hughes Medical Institute. "Laboratories at Yale are making progress toward elucidating some of these paths."
De Camilli is senior author of the two new papers, which are published in the Journal of Cell Biology and Proceedings of the National Academy of Science (PNAS).
Previous studies have shown that mutations of the gene VPS13C cause rare cases of inherited Parkinson's or an increased risk of the disease. To better understand why, De Camilli and Karin Reinisch, the David W. Wallace Professor of Cell Biology and of Molecular Biophysics and Biochemistry, have investigated the mechanisms by which these mutations lead to dysfunction on a cellular level.
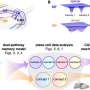
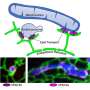
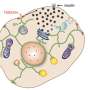
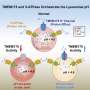
In 2018 they reported that VPS13C forms a bridge between two subcellular organelles—the endoplasmic reticulum and the lysosome. The endoplasmic reticulum is the organelle that regulates the synthesis of most phospholipids, fatty molecules that are essential for building cell membranes. The lysosome acts as a cell's digestive system. They also showed that VPS13C can transport lipids, suggesting that it may form a conduit for the traffic of lipid between these two organelles.
One of the new papers from De Camilli's lab demonstrates that the lack of VPS13C affects the lipid composition and properties of lysosomes. Moreover, they found that in a human cell line these perturbations activate an innate immunity. Such activation, if occurring in brain tissue, would trigger neuroinflammation, a process implicated in Parkinson's by several recent studies.
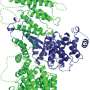
The second paper from De Camilli's lab uses state-of-the-art cryo-electron tomography techniques to reveal the architecture of this protein in its native environment supporting a bridge model of lipid transport. Jun Liu, a professor of microbial pathogenesis at Yale, is co-corresponding author of this study.
Understanding these fine-grained molecular details will be crucial in understanding at least one of the roads that lead to Parkinson's disease and may help identify therapeutic targets to prevent, or slow, the disease, researchers say.
Yale's William Hancock-Cerutti is lead author of the paper appearing in the Journal of Cell biology and Shujun Cai is lead author of the paper published in PNAS.
More information: William Hancock-Cerutti et al, ER-lysosome lipid transfer protein VPS13C/PARK23 prevents aberrant mtDNA-dependent STING signaling, Journal of Cell Biology (2022). DOI: 10.1083/jcb.202106046
Shujun Cai et al, In situ architecture of the lipid transport protein VPS13C at ER–lysosome membrane contacts, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2203769119