Researchers reveal SERCA2a as a molecular link between insulin resistance and early diabetic cardiomyopathy
Diabetic cardiomyopathy (DCM) is a progressive complication associated with type 2 diabetes (T2D), causing deterioration of cardiac function in diabetic patients, independent of coronary artery disease, and hypertension. It is possible for DCM to appear at the onset of diabetes, and can progress to heat failure.
DCM is a complex disorder triggered by multiple factors, such as glucose and lipid toxicity, oxidative stress, and ionic disturbances. Previous studies have shown abnormalities in cardiomyocyte calcium ion homeostasis in DCM; however, the underlying mechanism of its occurrence as well as its role in the pathogenesis of DCM is not yet clear.
Sarcoplasmic reticulum/endoplasmic reticulum ATPase 2a (SERCA2a) is a calcium pump for calcium ion recycling in the sarcoplasmic reticulum in cardiomyocytes. It transports calcium ions from the cytosol to the lumen of the sarcoplasmic reticulum by consuming ATP.
It has been widely acknowledged that SERCA2a is essential for maintaining cardiomyocyte calcium homeostasis and cardiac function, and alterations in its activity and expression are closely related to the onset and progression of heart failure. A recent study reported that SERCA2a can be activated by the SPEG (striated muscle preferentially expressed protein kinase)-mediated phosphorylation at threonine 484 (Thr484).
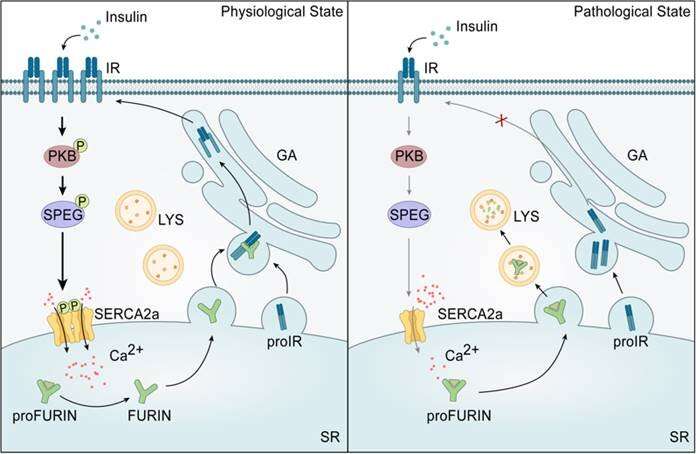
Another study revealed that insulin can activate the SPEG-SERCA2a pathway through the protein kinase B (PKB) in the heat, which in turn regulates cardiomyocyte calcium homeostasis. However, the activation mechanism of SERCA2a phosphorylation in vivo, as well as its role in the development of myocardial insulin resistance and DCM remains unclear.
A recent study published in Life Metabolism by Shuai Chen/Hongyu Wang at Nanjing university answered the above questions. This study identified a bidirectional regulatory mechanism between myocardial insulin resistance and calcium homeostasis dysregulation in cardiomyocytes mediated by phosphorylation of SERCA2a-Thr484, revealing a molecular mechanism in the early pathogenesis of DCM.
Mechanistically, SERCA2a regulates the protein stability of FURIN, the precursor protein shear enzyme that controls insulin receptor maturation, by modulating sarcoplasmic reticulum calcium recycling, and thus affects insulin sensitivity in cardiac myocytes. In line with this, impaired phosphorylation of SERCA2a-Thr484 leads to the degradation of FURIN, and therefore deceases the mature form of insulin receptor, finally exacerbating myocardial insulin resistance.
Overall, this study demonstrated that impaired phosphorylation of SERCA2a plays a bidirectional role in myocardial insulin resistance, dysregulation of calcium homeostasis and in the early stages of DCM, providing potential molecular targets for early intervention in DCM.
More information: Chao Quan et al, Impaired SERCA2a phosphorylation causes diabetic cardiomyopathy through impinging on cardiac contractility and precursor protein processing, Life Metabolism (2022). DOI: 10.1093/lifemeta/loac013