Safety and efficacy of intraventicular immunovirotherapy with oncolytic HSV-1 for CNS cancers
Metastatic or leptomeningeal disease (LMD), the spreading of a brain cancer to the meninges, is a devastating cancer complication that does not respond well to conventional therapies. While researchers can directly inoculate some brain tumors with an engineered virus that selectively targets cancer cells, this invasive technique is not feasible for widespread or surgically inaccessible malignancies, such as in LMD.
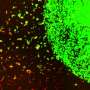
Researchers from Brigham and Women's Hospital and collaborators demonstrated that an alternative method of immunotherapy delivery, intraventricular (IVT) treatment, can be used to administer multiple therapeutic doses in mouse models of human metastatic medulloblastoma, prolonging survival while surmounting concerns about toxicity associated with the IVT method.
Researchers including Joshua Bernstock, MD, Ph.D., of the Brigham's Department of Neurosurgery and University of Alabama at Birmingham's Department of Pediatrics, previously treated pediatric cerebellar brain cancer with the immunotherapy tested in this study, oncolytic HSV (oHSV) G207, by directly inoculating tumors.
In this study, Bernstock and colleagues showed that administering protective pre-treatments and targeting the brain's ventricle system (cavities filled with cerebrospinal fluid) with low-dose oHSV G207 significantly extended survival in mice with a highly aggressive cancer, with tumor bioluminescence disappearing in G207-treated mice while it continued to increase in controls.
"We determined the cause of toxicity from intraventricular oHSV and established methods for mitigating toxicity to treat disseminated brain tumors in mice," Bernstock said. "Our data demonstrating the preclinical safety and efficacy of intraventricular G207 are highly translatable and support future clinical trials of this therapeutic approach."
The findings were published in Clinical Cancer Research.
More information: Kyung-Don Kang et al, Safety and efficacy of intraventricular immunovirotherapy with oncolytic HSV-1 for CNS cancers, Clinical Cancer Research (2022). DOI: 10.1158/1078-0432.CCR-22-1382