Experts explain treatment options for a common cancer complication
Experts provide the first framework for treating a common and life-threatening metabolic complication of cancer known as hypercalcemia of malignancy in the Endocrine Society's new Clinical Practice Guideline.
The guideline, titled "Treatment of Hypercalcemia of Malignancy in Adults: An Endocrine Society Clinical Practice Guideline," was published online and will appear in the March 2023 print issue of The Journal of Clinical Endocrinology & Metabolism (JCEM).
Estimates suggest hypercalcemia, or excess calcium in the blood, can occur in between 2 percent and 30 percent of cancer patients. Hypercalcemia of malignancy (HCM) is most frequently seen in breast, lung and renal cancer as well as multiple myeloma.
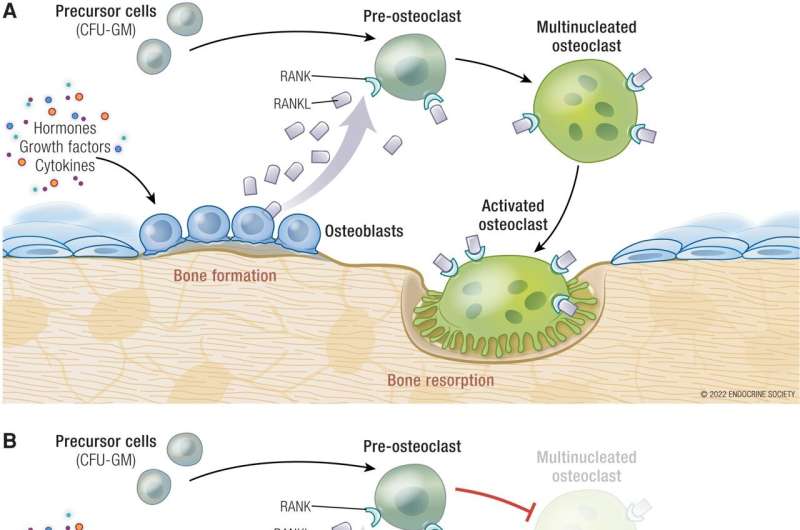
With the introduction of more powerful chemotherapy agents over several decades, HCM mortality rates have improved significantly. Treating the cancer is crucial for controlling the hypercalcemia and preventing it from recurring.
"Along with improved cancer treatments, we can treat HCM with medications to slow the resorption (breakdown) of bone and release of calcium into the blood," said Ghada El-Hajj Fuleihan, M.D., M.P.H., of the American University of Beirut in Beirut, Lebanon. El-Hajj Fuleihan is the chair of the panel that wrote the guideline.
"Health care providers currently have few evidence-based recommendations for which medications to use to treat HCM. Our guideline followed a rigorous process to examine the evidence available to date, and provides a clinical care workflow based on the severity and pathophysiology of HCM, taking into account contextual factors."
The expert panel recommended treating adults with HCM with either an intravenous (IV) bisphosphonate or denosumab medication. The panel suggested that treatment with denosumab is preferable to an IV bisphosphonate based on limited indirect evidence, which implied the higher potency of denosumab would translate into more favorable outcomes. In patients whose HCM recurs with IV bisphosphonate treatment, the panel suggested treatment with denosumab.
In adults with severe HCM, the panel suggested a combination of calcitonin with either bisphosphonate or denosumab as the initial treatment. Calcitonin treatment should be limited to 48-72 hours due to diminishing response thereafter.
The panel also provided guidance for the treatment of HCM due to calcitriol-secreting tumors and parathyroid carcinoma.
The Society established its Clinical Practice Guideline Program to provide endocrinologists and other clinicians with evidence-based recommendations in the diagnosis, treatment, and management of endocrine-related conditions.
Each guideline is developed by a multidisciplinary panel of topic-related experts in the field. Guideline development panels rely on evidence-based reviews of the literature in the development of guideline recommendations.
More information: Ghada El-Hajj Fuleihan et al, Treatment of Hypercalcemia of Malignancy in Adults: An Endocrine Society Clinical Practice Guideline, The Journal of Clinical Endocrinology & Metabolism (2022). DOI: 10.1210/clinem/dgac621. academic.oup.com/jcem/advance- … inem/dgac621/6916871