Mouse study suggests new therapeutic strategy to reduce cardiovascular disease in cancer survivors
Researchers at Memorial Sloan Kettering Cancer Center (MSKCC) in New York have discovered that common cancer treatments, such as radiotherapy or anthracycline drugs, cause long-term damage to heart tissue by activating a key inflammatory signaling pathway. The study, published in the Journal of Experimental Medicine (JEM), suggests that inhibiting this pathway could reduce the chances of cancer survivors suffering heart disease later in life.
Many cancers are treated with radiation and/or drugs that kill tumor cells by causing breaks in their DNA. But these treatments also damage the DNA of the patient's healthy cells. As the survival rates of cancer patients continue to rise, the long-term consequences of this are an increasing area of concern.
For example, radiotherapy or a class of DNA-damaging drugs known as anthracyclines can have delayed, toxic effects on the heart, increasing the risk of developing cardiovascular diseases, including coronary artery disease or heart failure. One study found that the occurrence of cardiovascular disease is five times higher in long-term survivors of Hodgkin's lymphoma than it is in the general population.
"The mechanisms by which DNA damage leads to late tissue toxicity years after cancer treatment have been poorly understood," says Adam M. Schmitt, a radiation oncologist at MSKCC. "Identifying the pathogenic mechanisms of toxicity and early biomarkers of their activation would provide an opportunity to intervene with treatment to prevent toxicity."
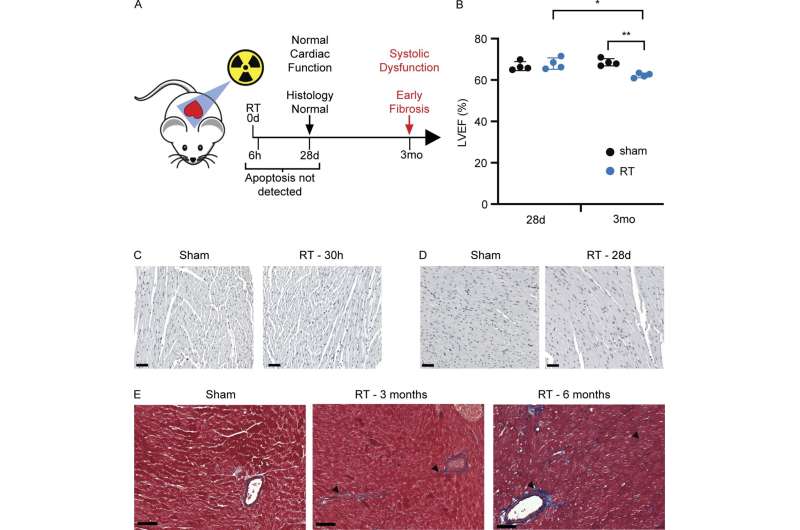
In their study, Schmitt and colleagues found that one month after mice were exposed to radiation or anthracyclines, a specific population of heart cells called fibroblasts activated a set of genes that promoted the recruitment of various immune cell types associated with pathological inflammation and tissue fibrosis. Within 3–6 months, the mice developed signs of cardiac dysfunction, and, by 12 months, many of them had died of heart failure.
The researchers determined that this pathological process is driven by an immune signaling pathway called the cGAS–STING pathway. This pathway usually promotes inflammation in response to DNA fragments derived from pathogenic bacteria or viruses, but, Schmitt and colleagues reasoned, it could also be activated by DNA fragments generated in response to radiation or anthracycline treatment.
Mice lacking either the cGAS or STING proteins were protected from the toxic side effects of DNA-damaging cancer treatments. They showed no signs of cardiac inflammation, maintained normal heart function, and were still alive a year after treatment. A small molecule inhibitor of the STING protein also protected mice from the toxic effects of radiotherapy or anthracyclines.
By studying breast cancer patients treated with anthracyclines, Schmitt and colleagues found evidence that the cGAS–STING pathway may play a similarly important role in human cardiac toxicity. One of the key inflammatory proteins induced by cGAS–STING signaling is CXCL10, and the researchers found that patients who showed the largest increases in CXCL10 levels after anthracycline treatment subsequently showed changes on echocardiograms associated with cardiac toxicity.
"Taken together, our data reveal that targeting of the cGAS–STING pathway holds great potential as a treatment to prevent cardiac complications of DNA-damaging cancer treatments," Schmitt says. "These data indicate that clinical trials utilizing cGAS–STING inhibitors to prevent cardiovascular disease following DNA-damaging cancer therapy are warranted."
More information: Achraf Shamseddine et al, Innate immune signaling drives late cardiac toxicity following DNA-damaging cancer therapies, Journal of Experimental Medicine (2022). DOI: 10.1084/jem.20220809