This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Portable cap can measure cognition with pulsed laser light
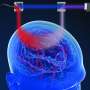
Measuring activity in the human brain remains one of the greatest challenges in science and medicine. Despite recent technological advances in areas such as imaging and nanoscience, researchers still struggle to accurately detect cognition. Currently, functional magnetic resonance imaging (MRI) is used to measure brain activity, but this method requires the patient to lie still in a large, noisy, and expensive apparatus. A portable and noninvasive method is needed to illuminate how the brain functions within a more natural setting while performing daily life activities.
In 2013, the National Institutes of Health launched an initiative to encourage more research into neuroscience by funding projects in key areas of the field. One such project is led by the Massachusetts General Hospital (MGH) Athinoula A. Martinos Center for Biomedical Imaging, in collaboration with MIT Lincoln Laboratory and Boston University, to develop a high-performance brain imaging method that can monitor cerebral blood flow with more accuracy than ever before. The brain regulates blood flow differently depending on what mental and physical tasks a person is doing. Accurately mapping cerebral blood flow with a portable system would give researchers insight into cognition.
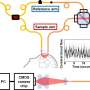
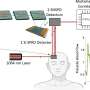
"This new method is called time-domain diffuse correlation spectroscopy (TD-DCS) and it works by transmitting laser light to and from the brain using fiber optics," says Jonathan Richardson, a research team member from Lincoln Laboratory's Advanced Imager Technology Group. The method will be integrated into a system resembling a cap that has 64 transmission and 192 receive points that are organized into groups called optodes, spaced 1 centimeter apart to cover nearly the entire scalp. "The light diffuses from the transmitter of each optode, bounces off of hemoglobin in red blood cells, and returns to several of the surrounding receivers."
Blood cells are constantly moving, and the faster they move, the more rapidly the intensity of the returning light signal will fluctuate. Researchers can use the rate of that fluctuation to measure blood flow velocity.
Early on in the program, the team worked to optimize the wavelength of light being used for the pulses. Tissue and blood absorb and scatter light differently at different wavelengths. These effects can swallow a light signal such that nothing bounces back to the receivers. Through modeling and measurements, they determined that a 1,064-nanometer laser could safely deliver almost 11 times more photons and reach a 25 percent deeper region of the brain than the shorter wavelengths that are used currently. In addition, a 1,064-nanometer laser is readily produced by commercial pulsed fiber laser technology.
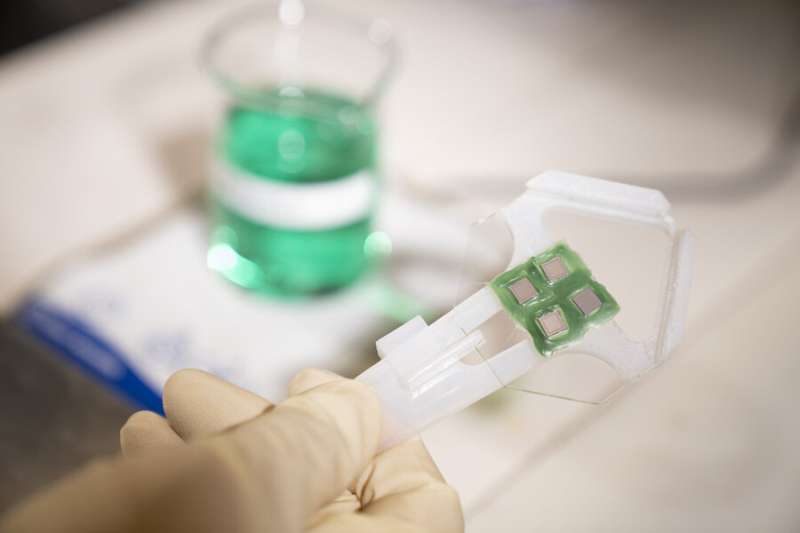
To make the receivers sensitive to faint light signals returning from deep in the brain, the team used a custom detector technology, developed at Lincoln Laboratory, called Geiger-mode avalanche photodiodes (GmAPDs).

"The GmAPD is a device that can give a fast electrical pulse in response to a single photon," says researcher Brian Aull. "We can detect that pulse and measure its time of occurrence digitally, which makes the detector exquisitely sensitive. We need that because most of the light scatters off in random directions and only a fraction of it scatters in the right direction to reach the detector."
Twenty years in the making, these GmAPDs have been involved in many critical programs at Lincoln Laboratory. This project is the first medical application of GmAPDs, which are coupled with a novel readout integrated circuit (ROIC) that was designed specifically for this use.
"After many years of development and demonstration for astronomy and national security applications, we are pleased to see our detector technology make an impact in medicine," says Erik Duerr, leader of the Advanced Imager Technology Group.
The GmAPD technology also addresses the issue of irrelevant returning light signals—in particular, those that bounce off cells in the scalp rather than blood in the brain—that can confound results.
"They are gated," says Aull, "meaning they can be turned on only during selected time intervals." Photons bouncing off of the scalp will return to the optodes more quickly than those coming from deeper in the brain. "By using a delayed turn-on, the system can ignore these early photons."
So far, the team successfully demonstrated TD-DCS at 1,064 nanometers in human subjects using individual commercial detectors. They are now focused on implementing and testing the ROIC and GmAPD integrated detector. In 2024, they plan to transition the system to the MGH team, who will then integrate it with their laser system.
"This technology has immediate clinical relevance to the diagnosis and tracking of traumatic brain injuries and can monitor brain perfusion during field-forward trauma care," says Richardson. "In the longer term, we hope this technology can assist in treatment of psychiatric conditions such as post-traumatic stress disorder, depression, and suicidality among soldiers as well."
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.