This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Psilocybin and mental health

Humans have long capitalized on the metabolic products of fungi for medical purposes (e.g., penicillin). Psilocybin is a fungal metabolite that has attracted increased attention from media outlets, clinicians and researchers in recent years. Naturally found in a multitude of mushroom species (colloquially known as "magic mushrooms"), the compound interacts with serotonin receptors in the brain to trigger various psychedelic effects.
A growing body of research suggests that, when administered in controlled conditions with supportive therapy, the compound may be useful for treating various psychiatric disorders, like depression. However, there are questions surrounding psilocybin's mechanism of action, as well as stigma, funding and regulatory hurdles, that must be addressed before psilocybin could be adopted for regular therapeutic use.
What is psilocybin and how does it work?
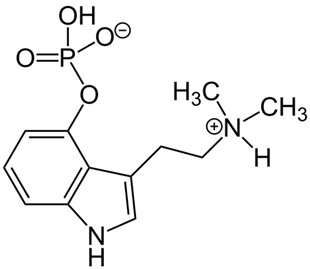
Psilocybin is naturally produced by over 200 species of basidiomycetes fungi—a phylum of filamentous fungi—collectively known as psilocybin mushrooms. These mushrooms come in all shapes and sizes and are found around the world. Psilocybin can also be synthetically produced and administered in standardized dosages, which is generally the form used in clinical settings.
Psilocybin belongs to a class of serotonergic psychedelics that includes lysergic acid diethylamide (LSD) and mescaline. Depending on the person and dosage, psilocybin can cause euphoria, changes in perception, perceived spiritual experiences and feelings of detachment, among other effects.
These effects are not due to psilocybin itself—rather, upon ingestion, it is metabolized to produce psilocin, the active form of the compound. Psilocin activates serotonin 5-HT2a receptors on cortical pyramidal cells in the brain. These cells are "the major computational units of the brain, which is where everything comes together. [They help us] put together our picture of what reality is," said David Nichols, Ph.D., a professor emeritus from Purdue University School of Pharmacy, who has been studying psychedelics since the late 1960s. By activating these receptors, psilocin alters brain metabolic activity and neuronal connectivity in ways that influence cognitive function and perception.
The history of psilocybin research: A winding road
Despite being used by people in Central America (and possibly other regions) for centuries, psychedelic mushrooms largely existed outside of Western societies until the 1950s, when an American mycologist, R. Gordon Wasson, took a trip to Huautla de Jiménez in Oaxaca, Mexico. There, he participated in a ritual using psychedelic mushrooms—the experience was captured in a 1957 article in Life magazine titled "Seeking the Magic Mushroom," which introduced the fungi to a broad audience.
That same year, Albert Hofmann, a chemist at the Swiss pharmaceutical company, Sandoz Pharmaceuticals, isolated psilocybin from a sample of dried Psilocybe mexicana mushrooms and identified it as the psychedelic "magic" in the mushrooms. As a result, Sandoz began manufacturing and distributing a pill containing psilocybin (Indocybin) for use as a research compound in psychiatric studies.
Trials exploring the efficacy of psilocybin were conducted throughout the 1960s. However, research on psilocybin bottomed out after the compound, along with other psychedelics like LSD and mescaline, was designated a Schedule 1 drug in 1970 by the United States Drug Enforcement Agency (DEA) as part of "the war on drugs." Studying Schedule 1 substances is possible, though doing so involves an arduous, expensive review and approvals process at the institutional and governmental levels. The stigmatization and regulatory barriers surrounding psilocybin slowed investigation of its therapeutic potential.
Recently, however, the gears have slowly started moving again. Seminal clinical studies in the mid-2000s paved the way for future endeavors funded by philanthropists and private organizations (e.g., the Heffter Research Institute, an organization founded by Nichols that helps design and fund psilocybin studies).
Now, guided by modern best practices for conducting psychedelic research, scientists at institutions across the U.S. are exploring psilocybin's applicability for multiple medical indications. Make no mistake: psilocybin is still a Schedule I compound, and it still is steeped in stigma. However, the more scientists study it, the more interest there is in continuing to do so. Sandeep Nayak, M.D., a psychiatrist and assistant professor at the John Hopkins University Center for Psychedelic and Consciousness Research, highlighted that "in the short [span] of a couple of months, we're getting lots and lots of studies out."
Nichols agreed that there's a tremendous amount of interest in psilocybin. A quick search on clinicaltrials.gov reveals over 130 trials using psilocybin that are actively underway or will be soon. "Five years ago, you wouldn't have found any," Nichols said.
The clinical potential of psilocybin
The influx of psilocybin research is fueled by compounding evidence that, when given under medical supervision in controlled conditions, psilocybin may be effective for managing various psychiatric conditions, including obsessive-compulsive disorder, alcohol use disorder and substance use disorders. It is also being investigated for smoking cessation, Alzheimer's disease, eating disorders and more.
While the list of conditions is growing, many studies so far have focused on psilocybin's potential for treating depression. Multiple clinical trials support psilocybin's ability to reduce depression symptom severity, including in people with treatment-resistant depression (i.e., failure to respond to at least 2 courses of traditional antidepressants, like SSRIs). A recent phase 2 double blind trial—the largest to date—showed that a single 25 mg dose of synthetic psilocybin administered in conjunction with psychotherapy led to significantly lower levels of depressive symptoms after 3 weeks compared to the control dose (1 mg). How? Researchers don't quite know yet.
"The only thing we know for sure is that [psychedelics, like psilocybin] activate serotonin receptors, but we really don't know their mechanism for producing an antidepressant—and not just antidepressant, but an antianxiety, antiaddictive—effect," Nichols explained. It may be that psilocybin alters neuronal connectivity in regions of the brain tied to rumination, responsiveness and emotion. In any case, COMPASS Pathways, the company that ran the trial, is beginning a phase 3 trial this year, with the goal of securing regulatory approval from the U.S. Food and Drug Administration (FDA) by the end of 2025.
The benefits of psilocybin treatment
When it comes to treating depression, Nichols pointed to psilocybin's speed and efficacy as 2 of its most attractive qualities. Psilocybin's effects are observable soon after administration (days) compared to standard antidepressants, which, when they work, can take weeks to fully kick in.
The antidepressant effects can also last for a while—from 4 weeks up to 6–12 months, depending on the study. While someone might have to take a standard SSRI daily for months or years, they might receive just 1 dose of psilocybin every few months, or even once per year. For Nayak, the "transdiagnostic" nature of psilocybin (i.e., ability to be used for multiple indications) is an additional plus.
The risks of psilocybin treatment
Still, Nayak emphasized that psilocybin does not work for everyone, and how well it works will vary from person-to-person. Moreover, in some cases, psilocybin can be harmful. "People that take [psychedelics] that shouldn't take them can have panic reactions and confusion, and it could precipitate psychosis in people who are predisposed to psychosis," such as those with schizophrenia or bipolar disorder, Nichols explained. Recruitment for clinical trials depends on careful screening to determine whether someone's personal and familial medical history may predispose them to adverse reactions.
Yet, even with careful screening, psilocybin is not perfect. In the COMPASS Pathways study, 77% of participants had headache, nausea, fatigue and dizziness, regardless of psilocybin dosage. A few participants reported suicidal ideation and self-injurious behavior which, though "common in treatment-resistant depression studies," according to the company, must be considered as research progresses.
While many of the above effects are not unique to psilocybin relative to traditional antidepressants, the stigma surrounding its recreational uses places it in a harsher light from a regulatory standpoint—according to Nayak, these types of outcomes could form a barrier to FDA approval. "I don't think it's [ever] going to be a situation where your doctor writes you a prescription for psilocybin," Nichols said. "I think it's always going to have a level of control for safety."
Psilocybin's path forward
Already, states like Colorado and Oregon have legalized therapeutic psilocybin use. There are, however, basic questions about psilocybin that lack solid answers. Clinicians are still learning how psilocybin works, who it works for and when and how often it should be used. Answering these questions will require completion of more clinical trials with a greater number of participants.
Obtaining funding for such studies is a challenge—the stigmatization of psilocybin has prompted governmental funding bodies to avoid projects involving the compound. However, Nichols thinks that, as trials demonstrating the potential benefits of psilocybin keep coming out, there may be a greater push and incentive for investment. He and Nayak believe this will depend, in part, on a "reeducation process" to help people disentangle historical connotations about psilocybin from what emerging scientific data show—that psilocybin-assisted therapy can, in some cases, be a beneficial thing.
Should psilocybin-assisted therapy be FDA-approved, both researchers highlighted that working out the therapy's scalability and affordability will be of utmost importance. In experimental trials, psilocybin-assisted therapy can cost tens of thousands of dollars per patient. Reaching a point where it is universally accessible (e.g., covered by insurance companies) will be key.
All of this takes time, but things are moving. "It's kind of like a snowball rolling downhill very, very slowly," Nichols said. "It's a work in progress."