This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Chemotherapy alters immune cell landscape in pancreatic cancer
Chemotherapy affects the ability of a patient's immune system to attack pancreatic tumors, a new study shows.
Led by researchers at NYU Langone Health and its Perlmutter Cancer Center, the work revolves around the immune system, which includes T cells designed to attack foreign organisms like viruses. To spare normal cells, the system uses "checkpoint" molecules like PD1 on T cell surfaces to turn off their attack when they receive the right signals.
The body also recognizes tumors as abnormal, but cancer cells hijack checkpoints to turn off immune responses. A prominent type of immunotherapy seeks to shut down checkpoints, thereby making cancer cells "visible" again to the immune system.
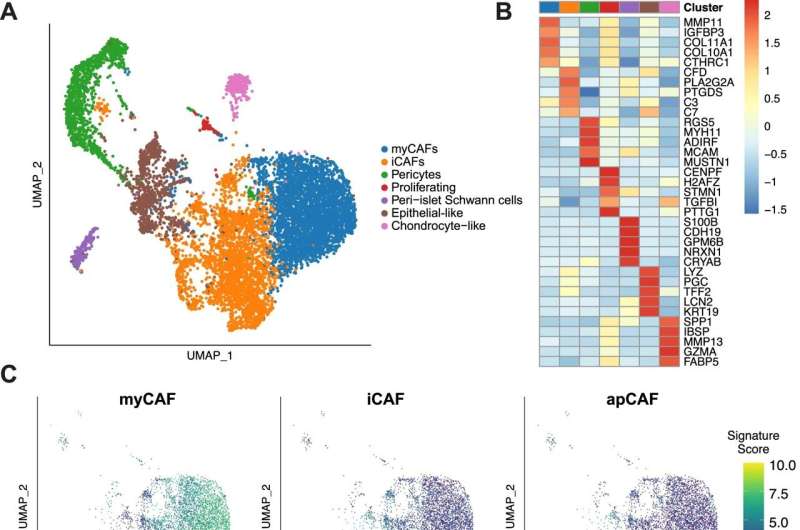
Published in the journal Nature Communications online Feb.13, the study analyzed more than 139,000 tumor cells collected from 27 patients with pancreatic ductal adenocarcinoma (PDAC), a hard to detect and treat cancer, with only 12% of patients surviving longer than five years after diagnosis. According to researchers, a strong immune response to tumors in the tissue surrounding them is critical to shrinking these tumors in the pancreas.
Among the new findings was a threefold decrease in the production of certain inhibitory checkpoint molecules when comparing 11 patients before chemotherapy to six others after it. Blocking these "off" signals, specifically PD1, is the goal of current immunotherapies designed to fight many cancers, but which have so far proven unsuccessful against PDAC, researchers say.
Importantly, the study showed that relative to each other, a second checkpoint, TIGIT, was the most common inhibitory checkpoint molecule in PDAC and 18 times more available for therapeutic targeting than PD1 before chemotherapy, but just five times more available after chemotherapy.
The study authors say that these findings warrant further study into whether immunotherapy focused on TIGIT might be more effective in pancreatic ductal adenocarcinoma than immunotherapies that target PD1, or programmed cell-death protein 1.
"Our study demonstrates how chemotherapy can have profound effects on the cellular landscape of the tumor microenvironment in pancreatic ductal adenocarcinoma," said study co-senior investigator Aristotelis Tsirigos, Ph.D., a professor in the Departments of Medicine and Pathology at NYU Grossman School of Medicine, and a member of Perlmutter.
"Importantly, our results suggest that chemotherapy may promote resistance to subsequent immunotherapy in pancreatic ductal adenocarcinoma," said study co-senior investigator Diane Simeone, MD, Laura and Isaac Perlmutter Professor of Surgery in the Department of Surgery at NYU Grossman School of Medicine.
"Further research is needed to determine if, as a result of this potential resistance, chemotherapy needs to be combined with immunotherapy at the start of treating this stubborn and often deadly form of cancer," added Simeone, who is also a professor in the Department of Pathology at NYU Grossman, and director of the Pancreatic Cancer Center.
Other changes following chemotherapy were seen in the number of other immune cells present, in how much they interacted with each other, as well as decreases in other cancer-associated immune cells (e.g., fibroblasts and macrophages) that would if left unchecked encourage cancer growth. However, the precise impact of these molecular changes on treatment, the researchers note, remains to be defined.
Tsirigos says additional experiments are underway to validate the findings in more patients. He notes that further research is also needed to evaluate whether similarly detailed cell analyses, through a technique called scRNA-seq, of the tumor microenvironment shortly after diagnosis could help guide future treatment decisions.
"As new technologies allow us to see what is happening inside patients at the cellular level, we can start to adjust our evaluations of immunotherapies and possibly how best to use them based on what is actually happening around tumors," added Tsirigos, who also serves as co-director of NYU Langone's Division of Precision Medicine.
More information: Gregor Werba et al, Single-cell RNA sequencing reveals the effects of chemotherapy on human pancreatic adenocarcinoma and its tumor microenvironment, Nature Communications (2023). DOI: 10.1038/s41467-023-36296-4