This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study: SARS-CoV-2 infection has no clear negative effects on human oocyte and early embryo development
Since December 2022, the nationwide control policy on the COVID-19 epidemic has been changed in China. Fertility and in vitro fertilization (IVF) centers are receiving increasing numbers of infected patients. However, there is still a lack of high-quality evidence on the effects of the virus on human oocytes and early-stage embryos. Previous studies have not found the virus in the follicular fluid of infected individuals.
However, SARS-CoV-2-associated receptors such as angiotensin-converting enzyme 2 (ACE2) and transmembrane serine protease (TMPRSS2) are co-expressed in gametes and fertilized eggs as well as in blastocyst ectodermal cells, and viral infection may cause immune response that is detrimental to embryonic development, so it is reasonable to suspect that infection may affect oocyte and early-stage embryo quality.
At some centers, the decision has been made to cancel cycles or simply freeze oocytes for infected patients due to safety concerns. Herein researchers established a prospective cohort study to assess the impact of COVID-19 on oocyte quality and embryo development.
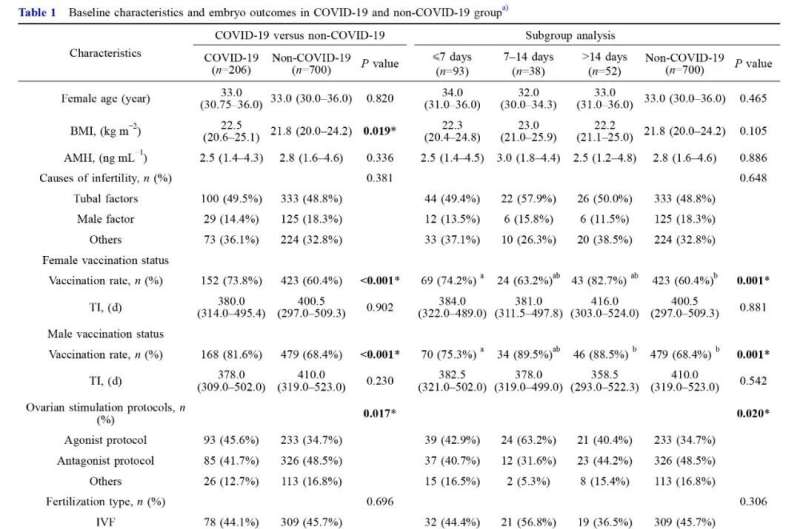
Valid data from a total of 906 couples were obtained from three reproductive centers in Shandong province and Shanghai municipality. Couples were divided into COVID-19 and non-COVID-19 groups depending on whether one member of the couples had been infected with SARS-CoV-2 before oocyte retrieval.
Based on the time interval from the infection of women to oocyte retrieval, the COVID-19 group was further subdivided into ≤7 days group, 7-14 days group and >14 days group. Oocyte quality and early embryo development indicators were followed-up and compared in each group to assess the effect of COVID-19.
Baseline characteristics were similar between COVID-19 and non-COVID-19 groups, with the exceptions of body mass index (BMI), vaccination status and ovarian stimulation protocols. There were no significant differences in oocyte-related outcomes and embryo development outcomes between COVID-19 group and non-COVID-19 group, except the number of bipronuclear (2PN) zygotes [6.0 (IQR 3.0–10.0) vs. 5.0 (IQR 2.0–8.0), P = 0.021].
Subgroup analysis showed ≤7 days group, >14 days group and non-COVID-19 group did not differ in oocyte and embryo laboratory outcomes with each other. However, for women who were infected 7-14 days before oocyte retrieval, more 2PN zygotes [8.5 (IQR 4.0-11.0) vs. 5.0 (IQR 2.0-8.0)] were obtained, along with a higher oocyte utilization rate [45.3% (IQR 34.6%-60.0%) vs. 35.0% (IQR 20.0%-53.8%)], than the non-COVID-19 group.
We further performed multivariable linear regression analysis by adjusting BMI, vaccination status and ovarian stimulation protocols. The adjusted results suggested that infection 7-14 days before oocyte retrieval was found to increase the number of oocytes retrieved, the number of 2PN zygotes, and the number of good-quality embryos. But infection within 7 days before oocyte retrieval diminished oocyte utilization rate.
The study also investigated the effect of COVID-19 on the female only and male only infected populations. The female only infected group had a higher number of 2PN compared to the non-COVID-19 group, with no significant differences in other indicators. In the male only infected group, there was no difference in outcomes compared to the non-COVID-19 group, but a reduction in progressive motility of sperm and good-quality embryos rate were identified in male only infected group despite there was no statistical significance.
Overall, the study suggested that COVID-19 did not have a clear negative effect on oocyte quality or embryo development. However, the pros and cons should be fully weighed in women with acute infection. This study provided favorable evidence for the successful implementation of ART during epidemic. It is hoped that this will inform clinical work and strengthen the confidence of patients in receiving ART treatment during COVID-19 epidemic.
Owing to the limitation of this study having a short follow-up and small sample size, the effects of COVID-19 on pregnancy and live birth are not clear yet. Researchers will further trace long-term pregnancy outcomes as well as the health of off-spring in this prospective cohort study.
More information: Xiaolei Chen et al, The effect of SARS-CoV-2 infection on human embryo early development: a multicenter prospective cohort study, Science China Life Sciences (2023). DOI: 10.1007/s11427-023-2291-0





















