This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New study reveals a possible cause of inflammation in Alzheimer's disease
Alzheimer's disease is characterized by the accumulation of plaques of the amyloid-β protein, chronic inflammation and impaired neuronal function in the brain. The most significant genetic risk factor for the disease is apoE4, a variant of apolipoprotein E, which is known for, among other things, advancing the onset of the disease. While more than half of all individuals with Alzheimer's disease carry this variant, the exact effect of apoE4 on the development of the disease has remained unknown.
A study recently completed at the University of Helsinki has identified a more accurate link between the apoE4 gene and the part of the human body's immune system that underlies, among other things, Alzheimer's disease. This is known as the complement system, and it contributes to the destruction of foreign cells and easily triggers inflammatory responses in the body.
The study, published in May in EMBO Reports, was carried out both by using experimental cell culture models and investigating brain biopsy samples from patients with idiopathic normal pressure hydrocephalus (iNPH) syndrome, in cooperation with the University of Eastern Finland, Kuopio University Hospital and international research organizations.
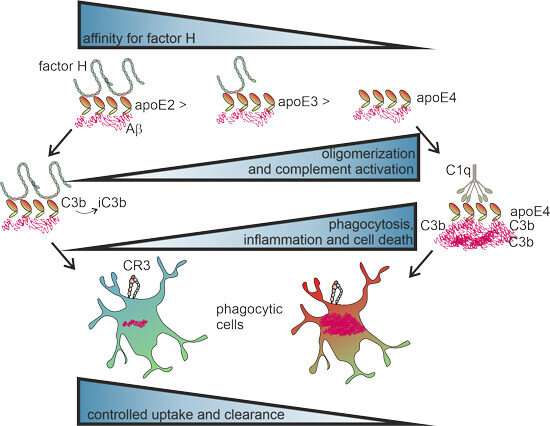
"We found that apoE4 poorly binds factor H, a regulatory factor of immunity. The factor H molecule is crucial in preventing complement-mediated inflammation," says Karita Haapasalo, the principal investigator of the study leading the Inflammation and Infections research group at the University of Helsinki.
"Usually, apoE binds factor H to the amyloid-β aggregates in the brain, thus reducing local inflammation. But apoE4 does not," she points out. This results in the accumulation of harmful amyloid-β aggregates and inflammation in the brain.
According to Haapasalo, binding factor H to apoE4 could present a potential solution to preventing changes in the brain that lead to the onset of Alzheimer's disease. Further research will soon be conducted to look for such a bridging molecule.
Understanding the disease mechanism is key to identifying better therapies
The amyloid-β aggregates associated with Alzheimer's disease begin to form in the brain decades before the diagnosis of the memory disorder. Since the mechanism underlying these changes has not been sufficiently well known, drug development has focused on stopping or slowing down changes that have already taken place.
"The drugs currently in use do not prevent the onset of the disease itself," Haapasalo confirms.
Alzheimer's disease is a slowly progressing memory disorder with more than 10 million new cases every year. As the population ages, the number of patients with the disease will rise considerably. In the future, Alzheimer's disease will increase human suffering, as well as the burden on public health and the economy.
"Determining the molecular mechanisms that affect the onset of Alzheimer's disease is important for developing curative drugs and therapies in the future."
More information: Larisa Chernyaeva et al, Reduced binding of apoE4 to complement factor H promotes amyloid‐β oligomerization and neuroinflammation, EMBO reports (2023). DOI: 10.15252/embr.202256467