This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Fruit flies stay sharp by flipping a metabolic switch in the brain

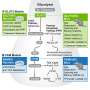
A new study led by scientists from TUD Dresden University of Technology reveals that the cells in the fruit fly brain possess a remarkable ability to shift their energy production to fats and send signals to the body's other organs, prompting them to start delivering lipids from fat stores to the brain during periods of starvation. The findings are published in the journal Nature Communications.
The brain, being one of the body's most energy-demanding organs, typically relies on sugar as its primary fuel source. However, during starvation, the brain can adapt and utilize alternative fuels, such as ketone bodies derived from stored fat. The question of whether brain cells can solely rely on externally obtained fuel or directly utilize fat has long puzzled scientists.
Metabolic switch
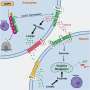
Led by Prof. Stefanie Schirmeier from the Faculty of Biology and Dr. Marko Brankatschk from the Biotechnology Center (BIOTEC) at TU Dresden, the research team used a comprehensive range of methods, including genetic manipulation, molecular biology, lipid analysis, and behavioral studies, to demonstrate how fruit fly brain cells can switch to fats to generate alternative neuronal fuel and safeguard against neurodegeneration.
"While neurons often come to mind when we think of the brain, there are other cells in the brain, called glia. They play vital roles in supporting and maintaining neurons. Our study shows that these glial cells respond to sugar shortage by activating fat utilization. In fruit flies, these cells utilize fats stored in lipid droplets or absorb lipids from circulation to produce ketones for the neurons to consume. This switch is crucial for the fly's survival," explains Dr. Marko Brankatschk, research group leader at BIOTEC.
Metabolic sensor
The study also revealed that glial cells act as messengers, signaling the body about the brain's energy shortage. "Our findings suggest that the metabolic switch in glial cells acts as a trigger, initiating a communication cascade that alerts the rest of the body to the challenging metabolic situation in the brain. As a result, the body's fat-storing organs mobilize reserves to sustain the brain's energy supply. It remains to be seen if a similar mechanism exists in the human brain," explains Prof. Schirmeier.
"Our work relies on Drosophila melanogaster, the common fruit fly, which has long served as a valuable model organism for studying development and disease. Our study demonstrates its potential to unlock insights into complex metabolic changes and their biological consequences," concludes Prof. Schirmeier.
The research conducted by TU Dresden scientists holds the potential to expand our understanding of brain metabolism and highlights the significance of studying simple model organisms to discover the intricate mechanisms underlying complex biological processes.
More information: Ellen McMullen et al, Glycolytically impaired Drosophila glial cells fuel neural metabolism via β-oxidation, Nature Communications (2023). DOI: 10.1038/s41467-023-38813-x