This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
COVID-19 mutations and their links to excessive immune responses
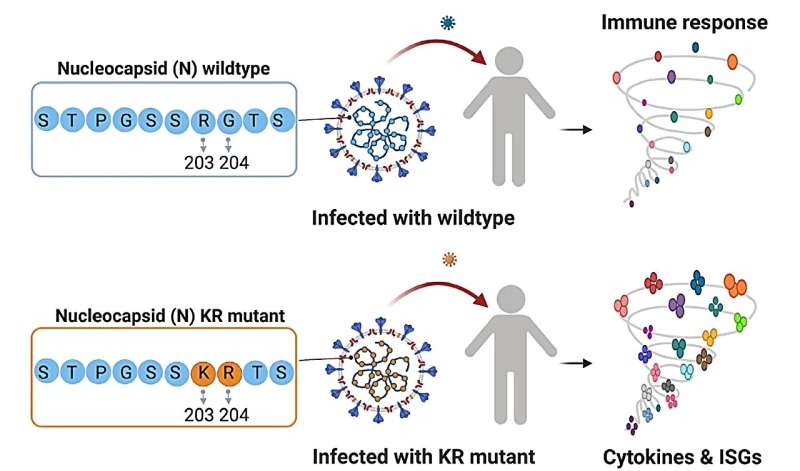
Mutations on a key SARS-CoV-2 viral protein are linked to extreme immune responses and could offer a novel vaccine target.
Since SARS-CoV-2 first appeared, scientists have been examining the structure of the virus for clues as to how it causes such diverse reactions in the human body. Now, KAUST researchers and scientists across Saudi Arabia have demonstrated how particular mutations on one SARS-CoV-2 protein are directly linked to extreme host immune responses.
For people who suffer a severe COVID-19 infection, the heightened immune response triggered by the virus can lead to systemic inflammation and, in the worst cases, a potentially deadly cytokine storm that can cause the patient's major organs to shut down.
"At the beginning of the pandemic, our laboratory initiated a collaborative research project on SARS-CoV-2 genomic epidemiology in Saudi Arabia as part of the KAUST Rapid Research Response Team (KAUST R3T)," says Muhammad Shuaib, epigenetics researcher and a member of the team led by Arnab Pain. Their previous study examined SARS-CoV-2 behavior and potential variations in its genetic code within the local population before vaccinations were available.

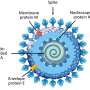
During this initial research, the team identified two adjacent, co-occurring amino acid mutations in the nucleocapsid protein (N-protein) of the SARS-CoV-2 virus. The N-protein is directly involved in viral replication within the host, meaning that it also interacts with the immune system. The N-protein mutations, known as 203K/204R (or KR), correlated with higher viral load and increased severity of COVID-19 symptoms.
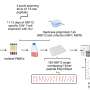
"Our findings raised questions about how the virus influences and interacts with the host immune system," says Shuaib. "For our current study, we combined genomics approaches to profile gene expression in patients infected with SARS-CoV-2 (with and without the KR mutation) and compared these results with healthy controls." The findings have now been published in the journal Genome Medicine.
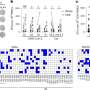
The team found that the expression of pro-inflammatory factors was significantly higher in people infected with the KR mutant SARS-CoV-2. These patients also had higher expression of interferon-stimulated genes, which trigger host immune defenses. Further, KR patients had an elevated neutrophil-to-lymphocyte ratio, suggesting their bodies were flooded with an excessive immune response. However, precisely how the KR mutations trigger these responses is still unclear. The transition from the original N-protein motif to the KR motif could hold clues.
"KR mutations might alter the protein's structural properties, changing its function and interactions," says Shuaib. "Or the mutations may impact viral replication or transcription processes, potentially leading to higher viral load and hyper-inflammatory responses."
The team is working closely with U.K. institutes to develop a viable animal model and examine these molecular mechanisms further. Notably, the N-protein is conserved throughout the coronavirus family, with interesting implications for future treatments.
"We might be able to use the mutant N-protein as a target for the development of universal vaccines," says Pain. "Such vaccines could offer broad protection not only against emerging SARS-CoV-2 variants, but also against future coronaviruses."
More information: Muhammad Shuaib et al, Impact of the SARS-CoV-2 nucleocapsid 203K/204R mutations on the inflammatory immune response in COVID-19 severity, Genome Medicine (2023). DOI: 10.1186/s13073-023-01208-0