This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
reputable news agency
proofread
Nobel in medicine goes to two scientists whose work enabled creation of mRNA vaccines against COVID-19

Two scientists won the Nobel Prize in medicine on Monday for discoveries that enabled the creation of mRNA vaccines against COVID-19 that were critical in slowing the pandemic—technology that's also being studied to fight cancer and other diseases.
Hungarian-American Katalin Karikó and American Drew Weissman were cited for contributing "to the unprecedented rate of vaccine development during one of the greatest threats to human health," according to the panel that awarded the prize in Stockholm.
WHAT IS THE NOBEL FOR?
The panel said the pair's "groundbreaking findings ... fundamentally changed our understanding of how mRNA interacts with our immune system."
Traditionally, making vaccines required growing viruses or pieces of viruses and then purifying them before next steps. The messenger RNA approach starts with a snippet of genetic code carrying instructions for making proteins. Pick the right virus protein to target, and the body turns into a mini vaccine factory.
But in early experiments with animals, simply injecting lab-grown mRNA triggered a reaction that usually destroyed it. Karikó, a professor at Szeged University in Hungary and an adjunct professor at the University of Pennsylvania, and Weissman, of the University of Pennsylvania, figured out a tiny modification to the building blocks of RNA that made it stealthy enough to slip past immune defenses.
Karikó, 68, is the 13th woman to win the Nobel Prize in medicine. She was a senior vice president at BioNTech, which partnered with Pfizer to make one of the COVID-19 vaccines. Karikó and Weissman, 64, met by chance in the 1990s while photocopying research papers, Karikó told The Associated Press.

WHY DO MRNA VACCINES MATTER?
Dr. Paul Hunter, a professor of medicine at Britain's University of East Anglia, described the mRNA vaccines made by BioNTech-Pfizer and Moderna Inc. as a "game changer" in shutting down the coronavirus pandemic, crediting the shots with saving millions of lives.
"We would likely only now be coming out of the depths of COVID without the mRNA vaccines," Hunter said.
John Tregoning, of Imperial College London, called Karikó "one of the most inspirational scientists I have met." Her work together with Weissmann "shows the importance of basic, fundamental research in the path to solutions to the most pressing societal needs," he said in a statement.
The duo's pivotal mRNA research was combined with two other earlier scientific discoveries to create the COVID-19 vaccines. Researchers in Canada had developed a fatty coating to help mRNA get inside cells to do its work. And studies with prior vaccines at the U.S. National Institutes of Health showed how to stabilize the coronavirus spike protein that the new mRNA shots needed to deliver.

Dr. Bharat Pankhania, an infectious diseases expert at Exeter University, predicted the technology used in the vaccines could be used to refine vaccines for other diseases like Ebola, malaria and dengue, and might also be used to create shots that immunize people against certain types of cancer or auto-immune diseases including lupus.
Peter Maybarduk, at the Washington advocacy group Public Citizen, welcomed the recognition of mRNA vaccines, but said the award should also be deeply embarrassing for Western countries.
"This is a technology that should have been available to all of humanity but it was almost exclusively available only in the richest countries in the world," he said, adding that much of the funding that led to the development of mRNA technology came from U.S. public funds.

HOW DID KARIKÓ AND WEISSMAN REACT?
"The future is just so incredible," Weissman said. "We've been thinking for years about everything that we could do with RNA, and now it's here."
Karikó said her husband was the first to pick up the early morning call, handing it to her to hear the news. She then watched the announcement to make sure she wasn't being pranked.
"I was very much surprised. But I am very happy."
Karikó said she was the one to break the news to Weissman, since she got in touch before the Nobel committee could reach him.
The two have collaborated for decades, with Karikó focusing on the RNA side and Weissman handling the immunology: "We educated each other," she said.
Before COVID-19, mRNA vaccines were already being tested for other diseases like Zika, influenza and rabies—but the pandemic brought more attention to this approach, Karikó said. It's also being tested for cancer immunotherapy.
-

Japan Prize 2022 laureate, American physician-scientist Drew Weissman poses with his trophy during the Japan Prize presentation ceremony Wednesday, April 13, 2022, in Tokyo. Two scientists have won the Nobel Prize in medicine for discoveries that enabled the development of mRNA vaccines against COVID-19. The award was given to Katalin Karikó and Drew Weissman. Karikó is a professor at Sagan’s University in Hungary and an adjunct professor at the University of Pennsylvania. Credit: AP Photo/Eugene Hoshiko, Pool, File -

Hungarian biochemist Katalin Kariko poses for a photo in Budapest, Hungary, Saturday, June 3, 2023. Two scientists have won the Nobel Prize in medicine for discoveries that enabled the development of mRNA vaccines against COVID-19. The award was given to Katalin Karikó and Drew Weissman. Karikó is a professor at Sagan’s University in Hungary and an adjunct professor at the University of Pennsylvania. Credit: Robert Hegedus/MTI via AP, FIle -

Hungarian biochemist Katalin Kariko poses for a photo in Budapest, Hungary, May 27, 2021. Two scientists have won the Nobel Prize in medicine on Monday, Oct. 2, 2023 for discoveries that enabled the development of mRNA vaccines against COVID-19. The award was given to Katalin Karikó and Drew Weissman. Karikó is a professor at Sagan’s University in Hungary and an adjunct professor at the University of Pennsylvania. Credit: Csilla Cseke/MTI via AP, File -

Nobel committee member Rickard Sandberg speaks during a press conference at the Karolinska Institute in Stockholm, Monday Oct. 2, 2023. The Nobel Prize in medicine has been awarded to Katalin Karikó and Drew Weissman for discoveries that enabled the development of effective mRNA vaccines against COVID-19. Credit: Jessica Gow/TT News Agency via AP -

Hungarian biochemist Katalin Kariko receives the highest state honour, the Grand Cross of the Hungarian Order of Saint Stephen from Hungarian President Katalin Novak, second right, in the Esztergom Basilica in Esztergom, northern Hungary, Sunday, Aug. 20, 2023. Two scientists have won the Nobel Prize in medicine on Monday, Oct. 2, 2023 for discoveries that enabled the development of mRNA vaccines against COVID-19. The award was given to Katalin Karikó and Drew Weissman. Karikó is a professor at Sagan’s University in Hungary and an adjunct professor at the University of Pennsylvania. Credit: Noemi Bruzak/MTI via AP, File -

Hungarian biochemist Katalin Kariko attends the Semmelweis Ignac Award in Budapest, Hungary, May 25, 2021. Two scientists have won the Nobel Prize in medicine on Monday, Oct. 2, 2023 for discoveries that enabled the development of mRNA vaccines against COVID-19. The award was given to Katalin Karikó and Drew Weissman. Karikó is a professor at Sagan’s University in Hungary and an adjunct professor at the University of Pennsylvania. Credit: (Zoltan Balogh/MTI via AP, File
"There was already clinical trials before COVID, but people were not aware," she said.
Karikó's family are no strangers to high honors. Her daughter, Susan Francia, is a double Olympic gold medalist in rowing, competing for the United States.
The prize carries a cash award of 11 million Swedish kronor ($1 million)—from a bequest left by the prize's creator, Swedish inventor Alfred Nobel. The laureates are invited to receive their awards at ceremonies on Dec. 10, the anniversary of Nobel's death.
Nobel announcements continue with the physics prize on Tuesday, chemistry on Wednesday and literature on Thursday. The Nobel Peace Prize will be announced Friday and the economics award on Oct. 9.
The Nobel Committee announcement:
The Nobel Assembly at Karolinska Institutet has today decided to award the 2023 Nobel Prize in Physiology or Medicine jointly to
Katalin Karikó and Drew Weissman
for their discoveries concerning nucleoside base modifications that enabled the development of effective mRNA vaccines against COVID-19.
The discoveries by the two Nobel Laureates were critical for developing effective mRNA vaccines against COVID-19 during the pandemic that began in early 2020. Through their groundbreaking findings, which have fundamentally changed our understanding of how mRNA interacts with our immune system, the laureates contributed to the unprecedented rate of vaccine development during one of the greatest threats to human health in modern times.
Vaccines before the pandemic
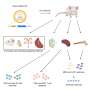
Vaccination stimulates the formation of an immune response to a particular pathogen. This gives the body a head start in the fight against disease in the event of a later exposure. Vaccines based on killed or weakened viruses have long been available, exemplified by the vaccines against polio, measles, and yellow fever. In 1951, Max Theiler was awarded the Nobel Prize in Physiology or Medicine for developing the yellow fever vaccine.
Thanks to the progress in molecular biology in recent decades, vaccines based on individual viral components, rather than whole viruses, have been developed. Parts of the viral genetic code, usually encoding proteins found on the virus surface, are used to make proteins that stimulate the formation of virus-blocking antibodies. Examples are the vaccines against the hepatitis B virus and human papillomavirus. Alternatively, parts of the viral genetic code can be moved to a harmless carrier virus, a "vector." This method is used in vaccines against the Ebola virus. When vector vaccines are injected, the selected viral protein is produced in our cells, stimulating an immune response against the targeted virus.
Producing whole virus-, protein- and vector-based vaccines requires large-scale cell culture. This resource-intensive process limits the possibilities for rapid vaccine production in response to outbreaks and pandemics. Therefore, researchers have long attempted to develop vaccine technologies independent of cell culture, but this proved challenging.
mRNA vaccines: A promising idea
In our cells, genetic information encoded in DNA is transferred to messenger RNA (mRNA), which is used as a template for protein production. During the 1980s, efficient methods for producing mRNA without cell culture were introduced, called in vitro transcription. This decisive step accelerated the development of molecular biology applications in several fields. Ideas of using mRNA technologies for vaccine and therapeutic purposes also took off, but roadblocks lay ahead. In vitro transcribed mRNA was considered unstable and challenging to deliver, requiring the development of sophisticated carrier lipid systems to encapsulate the mRNA. Moreover, in vitro-produced mRNA gave rise to inflammatory reactions. Enthusiasm for developing the mRNA technology for clinical purposes was, therefore, initially limited.
These obstacles did not discourage the Hungarian biochemist Katalin Karikó, who was devoted to developing methods to use mRNA for therapy. During the early 1990s, when she was an assistant professor at the University of Pennsylvania, she remained true to her vision of realizing mRNA as a therapeutic despite encountering difficulties in convincing research funders of the significance of her project. A new colleague of Karikó at her university was the immunologist Drew Weissman. He was interested in dendritic cells, which have important functions in immune surveillance and the activation of vaccine-induced immune responses. Spurred by new ideas, a fruitful collaboration between the two soon began, focusing on how different RNA types interact with the immune system.
The breakthrough
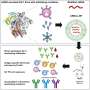
Karikó and Weissman noticed that dendritic cells recognize in vitro transcribed mRNA as a foreign substance, which leads to their activation and the release of inflammatory signaling molecules. They wondered why the in vitro transcribed mRNA was recognized as foreign while mRNA from mammalian cells did not give rise to the same reaction. Karikó and Weissman realized that some critical properties must distinguish the different types of mRNA.
RNA contains four bases, abbreviated A, U, G, and C, corresponding to A, T, G, and C in DNA, the letters of the genetic code. Karikó and Weissman knew that bases in RNA from mammalian cells are frequently chemically modified, while in vitro transcribed mRNA is not. They wondered if the absence of altered bases in the in vitro transcribed RNA could explain the unwanted inflammatory reaction. To investigate this, they produced different variants of mRNA, each with unique chemical alterations in their bases, which they delivered to dendritic cells. The results were striking: The inflammatory response was almost abolished when base modifications were included in the mRNA. This was a paradigm change in our understanding of how cells recognize and respond to different forms of mRNA. Karikó and Weissman immediately understood that their discovery had profound significance for using mRNA as therapy. These seminal results were published in 2005, fifteen years before the COVID-19 pandemic.
In further studies published in 2008 and 2010, Karikó and Weissman showed that the delivery of mRNA generated with base modifications markedly increased protein production compared to unmodified mRNA. The effect was due to the reduced activation of an enzyme that regulates protein production. Through their discoveries that base modifications both reduced inflammatory responses and increased protein production, Karikó and Weissman had eliminated critical obstacles on the way to clinical applications of mRNA.
mRNA vaccines realized their potential
Interest in mRNA technology began to pick up, and in 2010, several companies were working on developing the method. Vaccines against Zika virus and MERS-CoV were pursued; the latter is closely related to SARS-CoV-2. After the outbreak of the COVID-19 pandemic, two base-modified mRNA vaccines encoding the SARS-CoV-2 surface protein were developed at record speed. Protective effects of around 95% were reported, and both vaccines were approved as early as December 2020.
The impressive flexibility and speed with which mRNA vaccines can be developed pave the way for using the new platform also for vaccines against other infectious diseases. In the future, the technology may also be used to deliver therapeutic proteins and treat some cancer types.
Several other vaccines against SARS-CoV-2, based on different methodologies, were also rapidly introduced, and together, more than 13 billion COVID-19 vaccine doses have been given globally. The vaccines have saved millions of lives and prevented severe disease in many more, allowing societies to open and return to normal conditions. Through their fundamental discoveries of the importance of base modifications in mRNA, this year's Nobel laureates critically contributed to this transformative development during one of the biggest health crises of our time.
Key publications
Karikó, K., Buckstein, M., Ni, H. and Weissman, D. Suppression of RNA Recognition by Toll-like Receptors: The impact of nucleoside modification and the evolutionary origin of RNA. Immunity 23, 165–175 (2005).
Karikó, K., Muramatsu, H., Welsh, F.A., Ludwig, J., Kato, H., Akira, S. and Weissman, D. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther 16, 1833–1840 (2008).
Anderson, B.R., Muramatsu, H., Nallagatla, S.R., Bevilacqua, P.C., Sansing, L.H., Weissman, D. and Karikó, K. Incorporation of pseudouridine into mRNA enhances translation by diminishing PKR activation. Nucleic Acids Res. 38, 5884–5892 (2010).
Katalin Karikó was born in 1955 in Szolnok, Hungary. She received her Ph.D. from Szeged's University in 1982 and performed postdoctoral research at the Hungarian Academy of Sciences in Szeged until 1985. She then conducted postdoctoral research at Temple University, Philadelphia, and the University of Health Science, Bethesda. In 1989, she was appointed Assistant Professor at the University of Pennsylvania, where she remained until 2013. After that, she became vice president and later senior vice president at BioNTech RNA Pharmaceuticals. Since 2021, she has been a Professor at Szeged University and an Adjunct Professor at Perelman School of Medicine at the University of Pennsylvania.
Drew Weissman was born in 1959 in Lexington, Massachusetts, U.S.. He received his MD, Ph.D. degrees from Boston University in 1987. He did his clinical training at Beth Israel Deaconess Medical Center at Harvard Medical School and postdoctoral research at the National Institutes of Health. In 1997, Weissman established his research group at the Perelman School of Medicine at the University of Pennsylvania. He is the Roberts Family Professor in Vaccine Research and Director of the Penn Institute for RNA Innovations.
More information: Advanced information: www.nobelprize.org/prizes/medi … dvanced-information/
© 2023 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.