This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Structural study points the way to better malaria drugs
Structural insights into a potent antimalarial drug candidate's interaction with the malaria parasite Plasmodium falciparum have paved the way for drug-resistant malaria therapies, according to a new study by researchers at Weill Cornell Medicine and Van Andel Institute.
The antimalarial molecule, TDI-8304, is a new class of experimental therapeutics targeting the proteasome, an essential, multiprotein complex in P. falciparum cells. Two years ago, the researchers showed in a preclinical study that TDI-8304 potently kills malaria parasites at multiple stages of their life cycle and clears parasites in animal models of malarial infection.
In the new study, published in Nature Communications, the researchers used cryo-electron microscopy (cryo-EM) to show how TDI-8304 fastens to its proteasome target. The high-resolution imaging also revealed what happens to P. falciparum, the major cause of malaria deaths, when it mutates to become less sensitive to TDI-8304.
"This study gives us insights that should help in the development of new proteasome-inhibitor antimalarials that are not very susceptible to resistance," said study co-senior author Dr. Gang Lin, an associate professor of research in microbiology and immunology at Weill Cornell Medicine.
"Remarkably, the mutation that makes P. falciparum less sensitive to TDI-8304 makes it more sensitive to compounds that target a different part of the parasite proteasome," said co-senior author Dr. Huilin Li, professor and chair of the Department of Structural Biology at Van Andel Institute. "Our cryo-EM analysis illuminated how that 'collateral sensitivity' occurs."
The study's first author was Dr. Hao-Chi Hsu, a research scientist in Dr. Li's lab at Van Andel Institute. Dr. Laura Kirkman, an associate professor of medicine and microbiology and immunology at Weill Cornell Medicine, also collaborated on the study.
A need for new anti-malaria strategies
Malaria continues to be a major public health threat, with roughly 250 million cases per year and more than 600,000 deaths, principally in young children. Although malaria vaccines have been developed—the World Health Organization currently recommends two of them—their effectiveness and uptake in malaria-endemic regions are far from ideal.
The widespread use of antimalarial drugs also has led to the emergence of drug resistance in malaria parasites. Thus, new antimalarial drugs that are durably effective and available in pill form would be particularly useful.
In 2014, Dr. Lin and Dr. Kirkman began collaborating to develop molecules that inhibit the activity of the P. falciparum proteasome without affecting the human counterpart.
They began working with Weill Cornell Medicine's Enterprise Innovation team and ultimately began a collaboration with the Sanders Tri-Institutional Therapeutics Discovery Institute (Tri-I TDI), which partners with faculty at Weill Cornell Medicine, Memorial Sloan Kettering Cancer Center and The Rockefeller University to translate groundbreaking biological discoveries into small molecule or biologic therapeutics.
TDI-8304 emerged from that effort. In a 2021 study, Dr. Lin and colleagues showed in lab-dish and preclinical model tests that TDI-8304 is highly effective, even against P. falciparum strains that are resistant to first-line antimalarials called artemisinins.
However, their early work showed that P. falciparum can acquire mutations that reduce the parasite's susceptibility to molecules like TDI-8304, which primarily targets one subunit of the parasite proteasome.
Structural insights reveal strengths and weaknesses of drug candidate
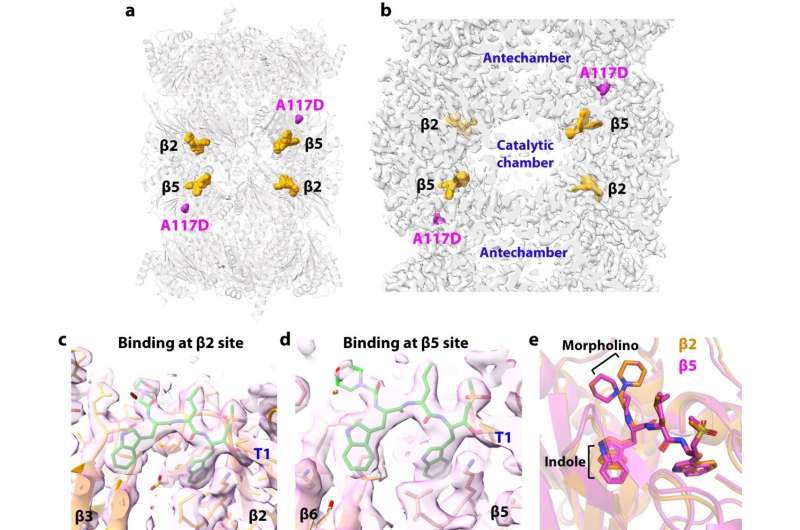
The main aim of the new study was to determine how TDI-8304 binds to the subunit, how that target differs on the human proteasome, and how the parasite's new mutation enables resistance. For this, Dr. Lin turned to his long-time collaborator, Dr. Li, a structural biology expert.
Cryo-EM studies of malaria parasite structures tend to be challenging due to the difficulty of growing the parasites in blood cells and enriching sufficient material. However, Dr. Li and his team succeeded in solving the TDI-8304/proteasome interactions at a very high resolution.
These models showed that TDI-8304 makes extensive connections to its target, whereas the new mutation, which "flips" a portion of the adjacent subunit, greatly weakens those connections.
Dr. Kirkman's prior work had shown that this mutation also somehow enhances the potency of a compound targeting a different part of the proteasome. In the new study, the resolution of the structure of this compound bound to the mutant proteasome clarified how this happens.
The researchers hope to use these findings to develop therapeutics that selectively and potently hit both these proteasome targets—a strategy that should delay the emergence of resistance. Drs. Lin and Li envision that such an approach could be combined with existing drugs to further minimize the risk of resistance.
More information: Hao-Chi Hsu et al, Structures revealing mechanisms of resistance and collateral sensitivity of Plasmodium falciparum to proteasome inhibitors, Nature Communications (2023). DOI: 10.1038/s41467-023-44077-2