This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Supercharging CAR-T cells for cancer treatment
At EPFL's School of Engineering, Professor Li Tang's Laboratory of Biomaterials for Immunoengineering has made significant strides in cancer treatment research. In laboratory settings, this innovative CAR-T therapy has consistently eradicated cancerous tumors in mouse models.
Separately, in ongoing clinical trials, eleven patients seemed to achieve complete remission using this treatment, marking a success rate of 100% to date. Notably, evidence from the lab study, published in Nature Biotechnology, suggests the therapy's long-term effectiveness and indicates that its fabrication may be both quicker and more cost-effective than current methods.
At its core, CAR-T therapy involves modifying T-cells to target and eliminate specific cancer cells. These modified T-cells are equipped with Chimeric Antigen Receptors (CARs) that allow them to recognize and latch onto cancer cells, marking a significant departure from traditional treatments.
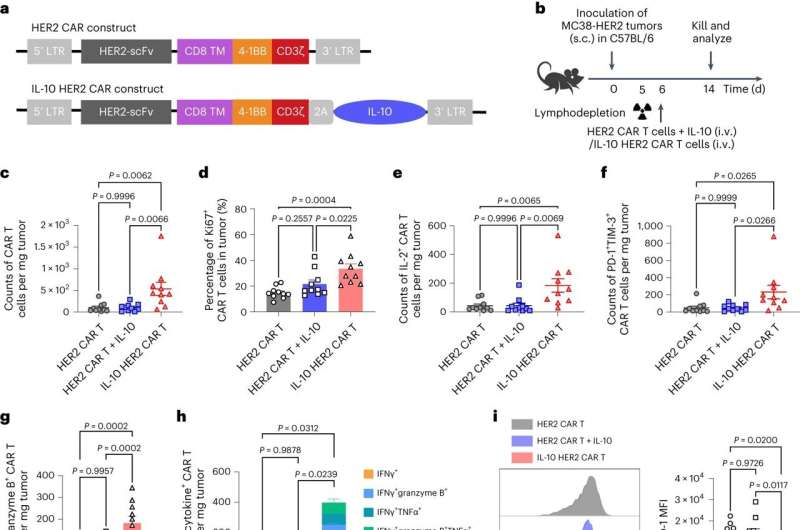
"We've added another layer to the CAR-T cell therapy by bioengineering a more robust, supercharged immune cell that is particularly efficient at targeting and destroying tumor cells," says Tang. The start-up Leman Biotech, co-founded by Tang and paper co-author Yugang Guo, aims to commercialize the treatment. The company has already garnered significant financial backing in its initial fundraising rounds.
Professor Li Tang's groundbreaking research adds another dimension to this innovative approach. Traditional CAR-T cells, while effective against liquid cancers, face challenges in solid tumors—the cells wear themselves out and ultimately failing to destroy the cancer fully.
Professor Tang's research introduces CAR-T cells that excrete the IL-10 molecule, which is then ingested by the modified T cells. In other words, the cell has been engineered to produce its own medicine to keep healthy in the tumor's hostile environment.
Surprisingly, the IL-10 molecule was traditionally viewed as an immune suppressant. But instead of inhibiting the immune response, Tang and his team have leveraged its unique metabolic reinforcement capabilities. This innovative twist bolsters the metabolism of the CAR-T cells. These metabolically armored treatments work immediately on existing tumors and have been shown to prevent future tumors from coming back.
Even after the reintroduction of tumor cells into the mouse models, the cells failed to establish themselves or show any malignancy. This underscores the lasting efficacy of the treatment, where the immune response remains vigilant and effectively neutralizes any renewed cancer threats. "The results in my lab are extremely exciting. We are convinced that this technology has the potential to save lives—as it has done so far with the 12 patients involved in our trial," says Tang.
While current CAR-T cell therapy has proven effective and several treatment options are currently available for leukemia and other liquid cancers, it remains extremely expensive: The cost of one treatment is upwards of $500k. In contrast, the costs of this future treatment could be significantly lowered due to the fact that only five percent of the traditional dose is necessary for full recovery.
Much of the costs come from fabricating relatively large amounts of these modified T-cells in expensive laboratory environments.
"A small amount of blood from a patient could provide already enough cells to prepare CAR-T cell therapy with our technology. The next day, you can already inject them back to the patient. It will be substantially less expensive and much faster to produce, saving more lives in the end," concludes Tang. Tang's team and Leman Biotech is currently working toward that goal.
More information: Yang Zhao et al, IL-10-expressing CAR T cells resist dysfunction and mediate durable clearance of solid tumors and metastases, Nature Biotechnology (2024). DOI: 10.1038/s41587-023-02060-8