This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New findings on the link between myotonic dystrophy 2 and autoimmune diseases
Myotonic dystrophy 2 (DM2) is a form of muscular dystrophy, a disease that leads to progressive muscle degeneration. It is caused by the expansion of a repetitive DNA sequence containing multiple CCTG bases in the CNBP gene. In general, the sequence of nucleobases in the DNA carries the genetic information.
Patients suffer from muscle weakness that is more pronounced in the area of the muscles close to the trunk, as well as sustained muscle stiffness and pain. Although DM2 occurs in roughly one out of 10,000 people in Germany, there are no targeted therapies.
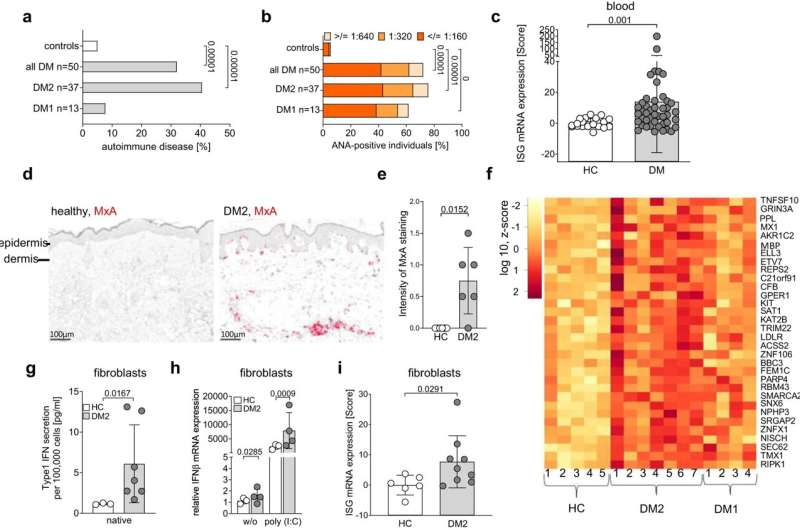
In initial studies, Prof. Claudia Günther and her team at the Carl Gustav Carus University Hospital at the Technical University of Dresden also observed that patients with DM2 suffer more from autoimmune diseases with an increased production of antibodies in the blood than the general population. However, the underlying mechanism for these symptoms was previously unknown.
Anti-viral response without a virus
In addition to the higher rates of autoimmunity, i.e. impaired tolerance to certain endogenous substances, Günther's team observed an unusual gene signature in cells derived from DM2 patients. "There were genes upregulated in the patient cells that are normally there to combat viruses," says first author Sarah Rösing, a doctoral student in Prof. Günther's Dresden research group.
"We quickly realized that this was an important discovery. While this type of immune response helps to combat viral infection, chronic activation is often connected to autoimmunity, so we really needed to understand where it comes from."
In order to elucidate the link between the expanded DNA repeats (CCTG)n found in DM2 patients, the antiviral response, and autoimmune diseases, scientists from Transregio 237 "Nucleic Acid Immunity" at the University Hospitals of Dresden and Bonn and the Cluster of Excellence ImmunoSensation2 at the University of Bonn collaboratively investigated the molecular signature in cells from patients with DM2.
DM2 cells are under chronic stress from junk proteins
In close collaboration, the researchers in Dresden and Bonn showed that in the cells of DM2 patients, the DNA repeats (CCTG)n are translated into toxic nonsense proteins. Their formation leads to a chronic form of stress on the endoplasmic reticulum (ER), a branched duct system as an extension of the nuclear membrane, which is where many proteins are folded. This ER Stress causes, in turn, chronic damage to the mitochondria, the energy powerhouse of the cells.
"Mitochondrial damage and ER stress in our cells are clear signals that something is wrong," comments Prof. Eva Bartok, one of the senior authors of the publication from the University Hospital Bonn and the University of Bonn, "This type of stress can definitely look like a viral infection and trigger an antiviral response."
These "stressed mitochondria" release small amounts of DNA within the cell, which are then recognized as a danger signal by the innate immune system. This is because cGAMP synthase (cGAS), an important innate immune receptor for DNA, recognizes this mitochondrial DNA and thus triggers an alarm state in the host by inducing the release of the key antiviral mediator, type I interferon.
"That was our 'aha' moment," says Prof. Günther, corresponding author of the publication. "We have identified a mechanism and a pathway that now open up new possibilities for targeted therapies of the disease."
These important findings offer new potential therapeutic targets to suppress the development of the autoimmune disease in these patients. "Our data provides an important rationale for inhibiting cGAS and the type I interferon pathway in myotonic dystrophy 2," comments Prof. Bartok.
"It is very exciting to see how the results of our basic research could potentially improve the care of these patients," concludes Prof. Günther.
The research is published in the journal Nature Communications.
More information: Sarah Rösing et al, Chronic endoplasmic reticulum stress in myotonic dystrophy type 2 promotes autoimmunity via mitochondrial DNA release, Nature Communications (2024). DOI: 10.1038/s41467-024-45535-1