This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Research reveals a process tumors use to induce immune suppressor cells and evade immunotherapy
Cleveland Clinic researchers recently published a study in Cell Reports that shines new light on a next-generation target of immunotherapy—the immune checkpoint protein VISTA.
Li Lily Wang, Ph.D., and a research team found a new, exploitable pathway for VISTA, which researchers know helps tumors develop "regulatory immune cells" called myeloid-derived suppressor cells (MDSCs). MDSCs prevent other immune cells that attack tumors, like T-cells, from functioning. Since VISTA's discovery in 2011, researchers have wondered exactly how the protein promotes MDSC development.
The hypothesis was that the regulatory function of VISTA was related to its ability to directly interact with T-cells and decrease their ability to fight diseases like infection and cancer. Dr. Wang's study proved that VISTA—and therefore immunotherapy resistance—operates through a completely different pathway.
Immunotherapies train the immune system to fight cancer, but proteins like VISTA can get in the way. Co-first author Amin Zakeri, Ph.D., explains that VISTA is an important "immune checkpoint protein" that poses a major obstacle to immunotherapies because it prevents our immune cells from properly responding to treatment.
Under healthy conditions, VISTA keeps immune systems in check and prevents attacks on healthy cells. During cancer, however, VISTA can do its job too well and prevents the body from successfully fighting off a tumor.
"Immunotherapies allow physicians to treat and manage cancers that could not previously be treated by chemotherapy or radiation therapy with a 20% to 30% rate of success," says Dr. Wang of Cleveland Clinic's Department of Translational Hematology & Oncology Research. "This is an incredible advancement, but to help the 70-80% of patients who fail to respond to this treatment, we must understand how tumors can evade the immune system."
The investigation into VISTA and MDSCs was led by three co-first authors: Dr. Zakeri, Keman Zhang, Ph.D., and Tyler Alban, Ph.D. The team showed that blocking VISTA in preclinical models reduced tumor size and increased the ability to respond to immunotherapy. They then took their study a step further to learn exactly how this response could occur.
"VISTA is involved in multiple pathways and works in a few different ways, so we expected to gain more insight into the already known pathways with our mechanistic studies," says Dr. Zhang, a research associate in Dr. Wang's lab. "Imagine our surprise when we found a totally new mechanism altogether."
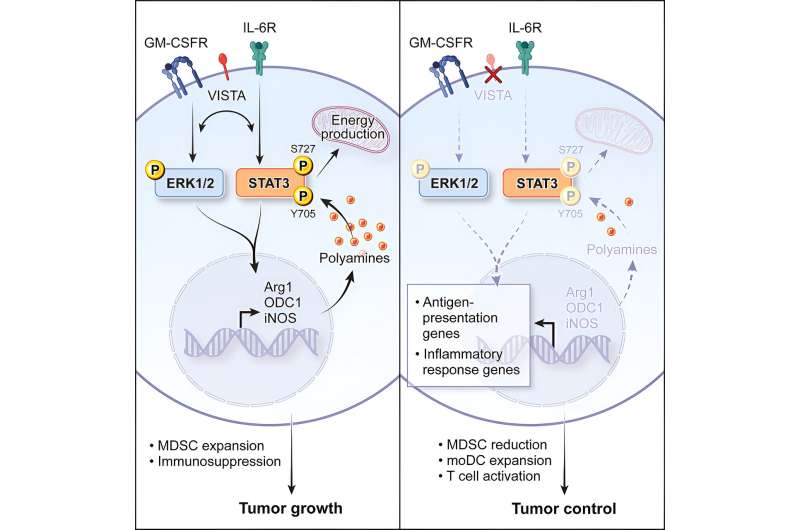
In the newly discovered pathway, VISTA is part of a feedback loop that promotes the activation of a gene called STAT3 (which encodes a protein of the same name) and the production of metabolites called polyamines. Polyamines help cells to grow, function, and replicate, but during cancer, they can accumulate and induce tumor-associated MDSC development, which helps the tumor resist immunotherapy.
There has been a focus in the biomedical research community on using STAT3 inhibitors as polyamine-blocking therapeutics, with limited success.
"STAT3 is a very attractive drug target for immunotherapy, but so far, the potential drug candidates that inhibit it have largely failed to make an impact due to toxicity," says Dr. Wang. "Our data raises the potential of influencing STAT3 and polyamine production indirectly by targeting VISTA instead."
Toxicity in other tissues isn't as much of a concern because the VISTA protein is disproportionately enriched in tumor-associated MDSCs, Dr. Wang added.
These implications are especially exciting because STAT3 has already been widely studied, says Dr. Alban. Dr. Alban works as a postdoctoral fellow at Cleveland Clinic's Center for Immunotherapy & Precision Immuno-Oncology. Connecting VISTA with a "known agent" like STAT3 will make follow-up studies and drug development much easier, Dr. Alban says, because scientists' abilities to make therapeutics are limited by their knowledge of the molecule they wish to target.
Research is currently underway to leverage the connection between STAT3 and VISTA in pursuit of an effective drug candidate. Dr. Wang says her team's study has a high translational impact on several human cancers, already tying VISTA expression with lower survival outcomes in endometrial cancer.
The team is developing the methods needed to determine what other cancer types use this mechanism in humans.
"There are many new collaborations and projects in the work to explore the mechanisms behind VISTA and its contribution to the tumor microenvironment," says Dr. Zakeri. "VISTA has opened a lot of new windows for immune and cancer research."
More information: Keman Zhang et al, VISTA promotes the metabolism and differentiation of myeloid-derived suppressor cells by STAT3 and polyamine-dependent mechanisms, Cell Reports (2024). DOI: 10.1016/j.celrep.2023.113661