This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Review summarizes the latest knowledge on type 2 diabetes
Diabetes is a widespread metabolic disorder affecting over 500 million adults worldwide. The most common form is type-2 diabetes (T2D), characterized by peripheral insulin resistance.
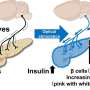
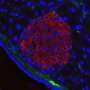
This implies that in patients with T2D, their peripheral tissues, such as muscle and fat cells, do not respond well to insulin, ultimately resulting in issues during the regulation of blood sugar levels. Another key feature of T2D is the progressive loss of function of b-cells in the pancreas, which are responsible for producing, storing, and releasing insulin.
T2D is generally manageable, with some patients relying on a strictly controlled diet, whereas others monitor their blood glucose levels and administer insulin as needed. Yet, poor management of the disease can lead to life-threatening consequences. To add to it, a definitive cure for T2D remains elusive.
Against this backdrop, a research team led by Professor Zhiguang Su from Sichuan University, China, recently decided to conduct an extensive literature review to summarize some of the latest knowledge pertaining to T2D. In their review, they touched upon the mechanisms that contribute to the failure of b-cells and potential therapeutic strategies.
The article was published in the Chinese Medical Journal.
The researchers first review the current understanding of the physiology of B-cells under normal conditions. They delve into the finer details of insulin production and secretion in these cells, exploring the regulated functions. Worth noting is the observation that insulin secretion is not a straightforward process regulated simply by sensing blood glucose levels; it rather resembles a complex chemical orchestration, influenced by various factors such as adenosine triphosphate (ATP), GABA neurotransmitters, and signaling between different pancreatic cell types.
The article then delves into the latest scientific progress aimed at understanding the origin and development of T2D, technically known as ' T2D pathogenesis.' As mentioned previously, insulin resistance is one of the hallmarks of T2D. It renders cells throughout the body unable to respond normally to insulin and coordinate a blood glucose-lowering response.
Interestingly, available evidence suggests that long-term insulin resistance cascades into the failure of b-cells. Explaining further, Prof. Su says, "Hyperglycemia, accompanied by obesity, particularly visceral adiposity, causes insulin resistance, and more insulin is needed to override the ineffectiveness of insulin. Pancreatic b-cells detect this requirement and adaptively augment insulin synthesis and secretion through compensatory expansion of their mass to restore glucose homeostasis."
Adding further, he says, "Ultimately, with increasing time, the number of b-cells as well as their secretory function progressively decline, and glucose homeostasis is impaired, eventually causing diabetes." The failure of b-cells is complex and is a result of a combination of mechanisms, including aging, genetics, oxidative stress, inflammation, and even the transformation of b-cells into other cell types.
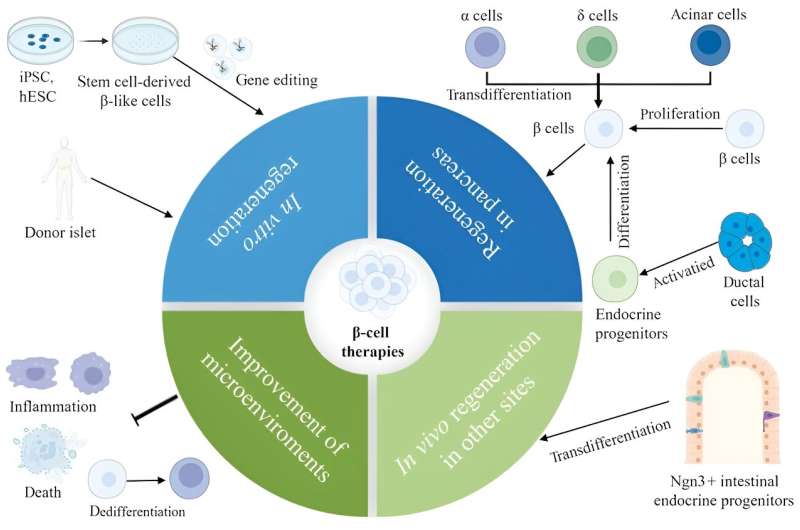
Finally, the review explores diverse therapeutic strategies aimed at replenishing and regenerating b-cells. As the review suggests, an optimal solution could involve stimulating the proliferation of one's b-cells through chemical signaling. Scientists have reported some success in using molecules produced by organs besides the pancreas and small-molecule drugs to induce B-cell proliferation. However, it is essential to note that these studies were conducted in mice, and thorough testing is required to determine the efficacy of these methods in humans.
Discussing these observations, an optimistic Professor Su says, "With the increasing appreciation of the mechanisms involved in promoting human b-cell proliferation and the development of high-throughput screening tools, it is anticipated that more small molecules and drugs expanding functional b-cell mass will be identified."
An alternative approach to address T2D is through pancreas transplants, either complete or partial. While promising a T2D cure, transplanted tissues face the challenge of immune system targeting, requiring recipients to take immunosuppressants, which can lead to a battery of new problems.
Another potential treatment for T2D could be the generation of b-cells using stem cells, presenting an immune system-friendly option. However, some important challenges will still remain to be addressed. For one, it appears not all b-cells are equal, and their proper functioning in the pancreas depends on having an appropriate balance of subpopulations. Moreover, so far, stem cell-derived B-cells do not perform as well as regular B-cells do.
In short, the review article presents a comprehensive summary of the recent advances in this field of research and will hopefully inspire researchers toward further research. With any luck, a cure for this common disorder might be within reach.
As Prof. Su remarks, "Although many challenges remain unanswered, we believe that b-cell regenerative therapies will represent a viable cure for diabetes in the not-too-distant future with the recognition of mechanisms responsible for b-cell development and mature endocrine cell plasticity, remarkable advances in improved protocols to generate b-cells from stem cells and single-cell studies."
More information: Daxin Cui et al, Pancreatic β-cell failure, clinical implications, and therapeutic strategies in type 2 diabetes, Chinese Medical Journal (2024). DOI: 10.1097/CM9.0000000000003034