This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Reticulon 3 deficiency ameliorates post-myocardial infarction heart failure by alleviating mitochondrial dysfunction
Heart failure (HF) after myocardial infarction (MI) is a global health issue with a high mortality rate. Multiple molecular mechanisms are involved in the development of HF after MI. However, interventions targeting these pathological processes alone remain clinically ineffective.
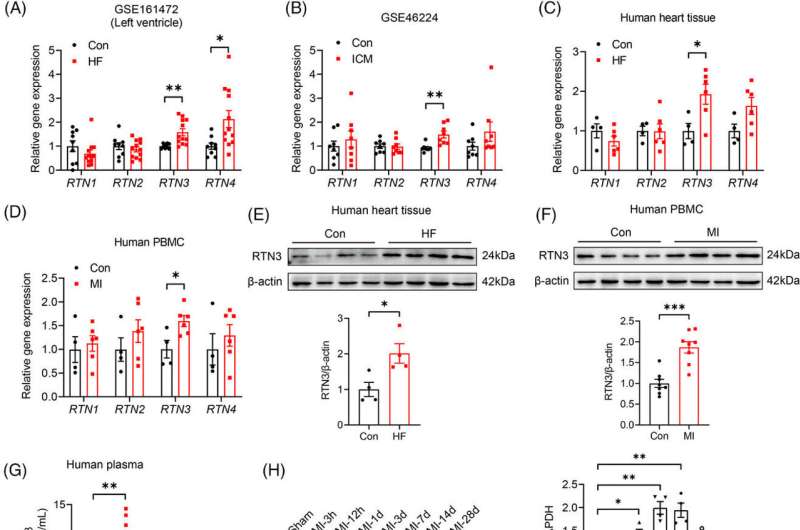
A study on this topic has been led by Dr. Yan Li and Dr. Mingming Zhang (Department of Cardiology, Tangdu Hospital, Air Force Medical University). The team found that reticulon 3 (RTN3), located in the endoplasmic reticulum (ER), is downregulated in the left ventricular tissue of patients with HF.
The findings are published in the journal MedComm.
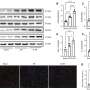
The researchers constructed a MI mouse model to further investigate the temporal and spatial specificities of RTN3 expression in the myocardium after MI. The results show that MI-induced increase in RTN3 level is mainly in the infarct region and infarct border zone rather than the remote zone.
Then, the team used gain- and loss-of-function approaches to investigate the role of RTN3 in HF after MI. Cardiomyocyte-specific RTN3 overexpression decreased systolic function in mice under physiological conditions and exacerbated the development of HF induced by MI. Conversely, RTN3 knockout alleviated cardiac dysfunction after MI.
"This result suggests that RTN3 may be a potential therapeutic target for HF after MI," says the first author of the paper, Bingchao Qi.
To explore the mechanism by which RTN3 knockout improves post-MI heart failure, the team performed co-immunoprecipitation (Co-IP) and mass spectrometry. Next, the Co-IP assay was respectively performed using the Flag antibody and HSPB1 antibody, confirming that RTN3 directly interacted with HSPB1.
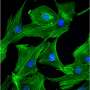
Further, RTN3 knockout significantly reduced HSPB1 expression in the ER and increased the content in the cytoplasm, while the overexpression of RTN3 resulted in the opposite effect. As expected, RTN3 knockout decreased TLR4 expression and increased PGC-1α expression in the myocardium post-MI, whereas AAV9-mediated RTN3 upregulation significantly increased TLR4 expression and decreased PGC-1α expression.
TLR4 can induce inflammatory responses after tissue injury. Surgical induction of MI resulted in the elevation of TLR4 expression and p-IκBα/IκBα level, whereas RTN3 knockout reversed the inflammatory signals. Activation of the TLR4/p-IκBα pathway is essential for NFκB phosphorylation and its translocation into the nucleus.
To verify this, the team extracted the nuclear components from cardiac tissues and demonstrated that RTN3 knockout reduced NFκB activation and nuclear translocation to a certain extent. The immunofluorescence assay revealed a significant decrease in MI-induced nuclear translocation of NFκB in the hearts of RTN3CKO mice.
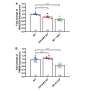
Additionally, PGC-1α is well-known as the key regulator of mitochondrial biogenesis. Therefore, the team determined the effect of RTN3 on critical genes of the mitochondrial ETC complexes. Downregulation of RTN3 increased the protein levels of mitochondrial complex-related genes after MI; however, its upregulation exerted an opposite effect.
Energy supply plays an essential role in the mitochondrial electron transport chain (ETC). The researchers observed that ATP production was increased in the hearts of RTN3 knockout mice and decreased in those of RTN3 overexpressing mice.
Owing to the mediating role of HSPB1 in RTN3-driven cardiac dysfunction, the team investigated whether pharmacological inhibition of HSPB1 could counteract the protective effects of RTN3 knockout on HF after MI. As expected, HSPB1 inhibition significantly inhibited the protective effect of RTN3 knockout on cardiac systolic function in mice after MI.
"Mitochondrial dysfunction and inflammation are important pathogenesis in several cardiovascular diseases, including MI and atherosclerosis. Therefore, targeting RTN3 using adeno-associated viruses or small molecule inhibitors may be a potential strategy for preventing and treating post-MI heart failure and other cardiovascular diseases," Qi says.
More information: Bingchao Qi et al, Reticulon 3 deficiency ameliorates post‐myocardial infarction heart failure by alleviating mitochondrial dysfunction and inflammation, MedComm (2024). DOI: 10.1002/mco2.503