This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
BRD7 research points to alternative insulin signaling pathway
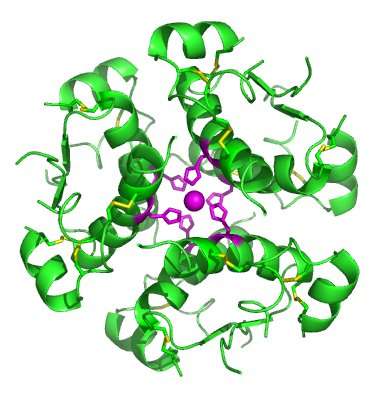
Bromodomain-containing protein 7 (BRD7) was initially identified as a tumor suppressor, but further research has shown it has a broader role in other cellular processes, including the remodeling of chromosomes and cell cycle progression.
Now, Boston Children's Division of Endocrinology researchers have discovered another purpose for BRD7: It seems to be involved in an alternative insulin signaling pathway, the existence of which had been speculated about for decades. Their findings, published in Journal of Endocrinology, could lead to new insights on previously unknown aspects of insulin signaling in obesity.
"The ultimate goal will be to find a way to reverse the type 2 diabetic features associated with obesity," says Sang Won Park, Ph.D., who was the corresponding author of this latest study on BRD7.
Looking at BRD7's function in glucose regulation
BRD7 has a key role in the regulation of glucose homeostasis—the balancing of glucose uptake in response to insulin and glucose production in the liver.
In 2014, Park and others found that the restoration of reduced BRD7 in the liver is sufficient to reestablish glucose homeostasis in obesity. Four years later, they looked at BRD7's participation in the insulin signaling pathway and its mechanistic function in the regulation of glucose homeostasis.
Building on that research, Park and others recently studied the interactive actions of BRD7 with insulin receptor substrates IRS-1 and IRS-2 (informally known as IRS1/2) in the livers of obese mice. IRS proteins were previously considered critical proteins required by the insulin receptor to transmit signals to downstream pathways.
Park's team—which included study co-first authors Junsik Lee and Yoo Kim, Ph.D., both of whom worked in her lab—had two goals: investigate the involvement of BRD7 as a downstream component of the insulin receptor (InsR) protein and examine the requirement of IRS1/2 for the action of BRD7.
They found that BRD7 interacts with the insulin receptor. This establishes BRD7 as an integral component of insulin signaling, Park says. Notably, they also discovered that IRS1/2 is not necessary for BRD7's regulation of glucose metabolism, particularly in the context of obesity. This finding indicates that BRD7 plays a role in an insulin signaling pathway independent of the pathway of the insulin receptor substrate-AKT axis.
Research continues to determine BRD7's role in potential treatment
Park and colleagues are now back to work. They hope to definitively prove that an alternative insulin signaling pathway exists. Further research will also hopefully illustrate how BRD7 is activated, either in the recognized signaling pathway or the alternative one posited by her team.
"We think it is related to the amount of energy and nutrients present in the body," Park says. "BRD7 somehow senses the availability of nutrition and blood glucose levels, but we're not exactly sure how that works."
Further research will include focusing on how BRD7 works within the insulin signaling mechanisms of obesity and non-obesity. Fully knowing BRD7's role could shape how a type 2 diabetes treatment is formed, Park says.
More information: Yoo Kim et al, BRD7 improves glucose homeostasis independent of IRS proteins, Journal of Endocrinology (2023). DOI: 10.1530/JOE-23-0119