This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study finds tyrosine kinase Csk promotes germinal center B cell survival and affinity maturation

The immune system strikes a fine balance by identifying and neutralizing disease-causing agents while carefully avoiding destruction of healthy tissues and cells. Now, researchers from Japan have shed new light on one of the processes that helps train immune cells to act only against genuine threats.
In a study published this month in the Journal of Experimental Medicine, researchers from Osaka University have revealed that a receptor protein with two distinct activities plays a key role in the development of an important subset of immune cells.
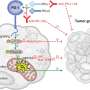
B cells, specialized cells in the immune system that recognize and "remember" foreign factors like bacteria, mature in the germinal center (GC) of immunity-related organs such as the spleen and the lymph node. Specific B cells that are particularly good at recognizing foreign substances are selected for and amplified through a process that involves B cell receptor (BCR) signaling. However, the mechanisms by which GC B cell positive selection takes place remain somewhat unclear.
"BCR is not only a signal transducer, but also mediates antigen presentation, a process that involves T cell–B cell interactions," says lead author of the study Takeshi Inoue. "This makes it challenging to determine each function separately during positive selection process."
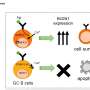
To tease apart the two functions of the BCR and explore their roles in GC B cell selection, the researchers used a mouse model lacking Csk, an inhibitor of BCR signaling. In these mice, BCR signaling was increased, but antigen presentation, the other function of the BCR, was unaffected.
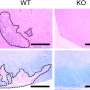
"The results were very clear," explains Tomohiro Kurosaki, senior author. "Deleting Csk decreased GC B cell survival and impaired their affinity maturation."
In addition, the mutated B cells showed mitochondrial dysfunction that was linked to higher levels of reactive oxygen species (ROS). The increase in ROS production led to B cell death, explaining why their survival rates were so much lower than those of unmutated cells.
"Our findings suggest that the Csk tyrosine kinase is a crucial regulator of GC BCR signaling that is required for B cell affinity maturation," says Inoue.
Given that appropriate ROS activity levels actually promote BCR signaling, the findings from this study suggest that whether ROS has both positive and negative effects on BCR signaling depends on the quantity of ROS that is present.
More information: Takeshi Inoue et al, Csk restrains BCR-mediated ROS production and contributes to germinal center selection and affinity maturation, Journal of Experimental Medicine (2024). DOI: 10.1084/jem.20231996