This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
CAR T cell therapy for advanced prostate cancer demonstrates positive results in Phase I clinical trial
Treating prostate cancer with immunotherapy is currently difficult to do. But results from a first in-human Phase I trial using a chimeric antigen receptor (CAR) T cell therapy developed by researchers from City of Hope, showed that patients with advanced prostate cancer can be treated safely with the cellular immunotherapy with promising therapeutic activity, according to the study published in Nature Medicine.
The trial treated 14 prostate stem cell antigen (PSCA)-positive patients who had metastatic castration resistant prostate cancer (mCRPC), which had spread beyond the prostate and no longer responded to hormone treatment, using CAR T cell therapy. More than 34,000 men with this type of prostate cancer die each year in the United States.
Saul Priceman, Ph.D., City of Hope associate professor, Department of Hematology & Hematopoietic Cell Transplantation, and team developed CAR T cells that target prostate stem cell antigen (PSCA) found to be highly expressed in prostate cancer.
The treatment took a patient's immune cells—known as T cells—from the bloodstream, and reprogrammed the cells in a laboratory with a CAR to recognize and attack the PSCA protein on the surface of cancer cells. CAR T cells were then infused back into the patient's system to destroy cancer cells.
"Prostate cancer has been called an immune desert—the tumor microenvironment is difficult to treat with immunotherapies because you don't get a lot of T cells inside the tumor," said Tanya Dorff, M.D., City of Hope section chief, Genitourinary Disease Program, and professor, Department of Medical Oncology & Therapeutics Research.
"It takes something really powerful to overcome that. Our study showed that City of Hope's CAR T cell therapy for prostate cancer could be a step closer to doing that."
"Our trial's major finding is that PSCA-directed CAR T cells are safe and do work against mCRPC," Priceman added. "This opens up the opportunity to continue to develop this type of cellular immunotherapy for these patients, who currently have no other effective treatment options."
The trial's goals were to examine the therapy's safety and dose-limiting toxicities, or side effects that limit the amount of treatment that can be administered, as well as preliminary data on the treatment's efficacy in patients.
The study's findings:
- Patients received a single infusion of 100 million CAR T cells without prior lymphodepletion chemotherapy, which is used routinely in blood cancers to improve the efficacy of CAR T cell treatment. Since this was a first-in-human CAR T cell trial, it was important to assess the safety of CAR T cells alone in patients.
- At that same CAR T cell dose and with lymphodepletion, there was a side effect of dose-limiting toxicity of cystitis, or irritation of the bladder. Dorff explained that PSCA is also found in the bladder so the CAR T cells most likely attacked the bladder cells, causing inflammation. Researchers then added a new cohort to the study using reduced lymphodepletion, which mitigated this toxicity.
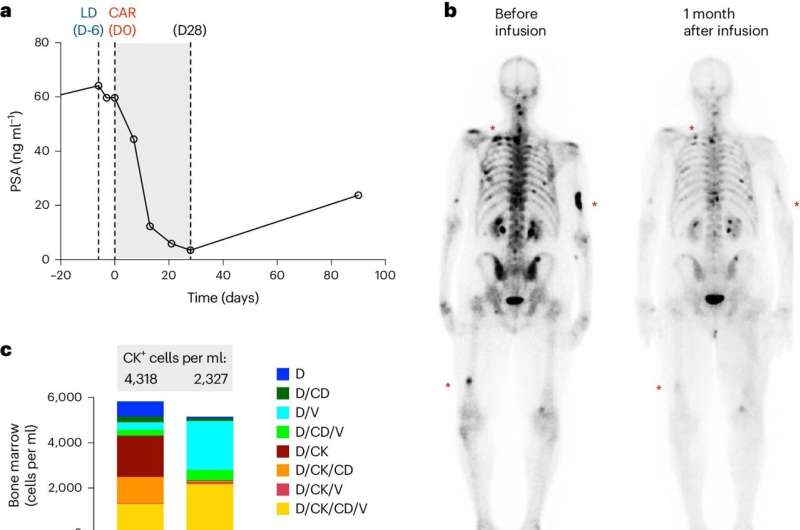
- Four out of 14 patients had declines in their PSA levels, which is a serum marker of disease progression in people with prostate cancer, including one patient with a significant decline. Imaging showed therapeutic responses in a subset of treated patients.
- Five out of 14 patients had mild or moderate cytokine release syndrome, which can be caused by a large, rapid release of cytokines into the blood from immune cells and is a common side effect after CAR T cell therapy. CRS was a treatable side effect.
- CAR T cells did not persist at high levels beyond the 28-day monitoring period, which limits the therapy's effectiveness. This presented a common challenge in the solid tumor CAR T cell field that researchers plan to address in a follow-up City of Hope trial using the therapy that is now open for enrollment.
One patient who had received several prior therapies responded well to the CAR T cell therapy. His PSA level decreased by 95% and cancer in his bones and soft tissue also declined. He experienced this positive response for approximately eight months.
"The patient's results were very encouraging, and we are deeply grateful for his participation in our study as well as other patients and their families," Dorff said. "We want to continue with this therapy and increase the amount of CAR T cells, and continue to carefully monitor for any health problems, as we think this can improve the therapy's effectiveness."
The Phase Ib trial using the PSCA-CAR T cell therapy in combination with radiation to enhance anti-tumor activity aims to enroll up to 24 patients.
More information: PSCA-CAR T cell therapy in metastatic castration-resistant prostate cancer: a phase 1 trial, Nature Medicine (2024). DOI: 10.1038/s41591-024-02979-8