This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New analysis offers most comprehensive roadmap to date for more targeted Alzheimer's research, drug discovery
From studying the human genome to analyzing the way proteins are encoded, or monitoring RNA expression, researchers are rapidly gaining a far richer understanding of the complex genetic and cellular mechanisms that underpin dementia. But there's a catch: While new technologies are revealing myriad avenues for Alzheimer's research, it's impossible to know in advance which research pathways will lead to effective treatments.
"We have countless potential targets, but we don't know which ones to aim at," said Greg Carter, the Bernard and Lusia Milch Endowed Chair at the Jackson Laboratory (JAX), who led the study. "Drug development is slow and costly, so to make use of these new insights, we need a way to prioritize them effectively."
Now, Carter and his colleagues at JAX—in collaboration with partners from Stanford University School of Medicine, Emory University, and Sage Bionetworks—are doing just that, offering the first comprehensive ranking of the relative role and significance of every gene and protein in the disease's development.
The research is reported in Alzheimer's & Dementia, in advance of the Alzheimer's Association International Conference on July 28, where the work will be presented.
"This is the most comprehensive study to date of Alzheimer's patients' brains," Carter said. "We're integrating research from multiple fields, including genetics and -omics, across the patient's lifespan, and at a much larger scale than has previously been possible."
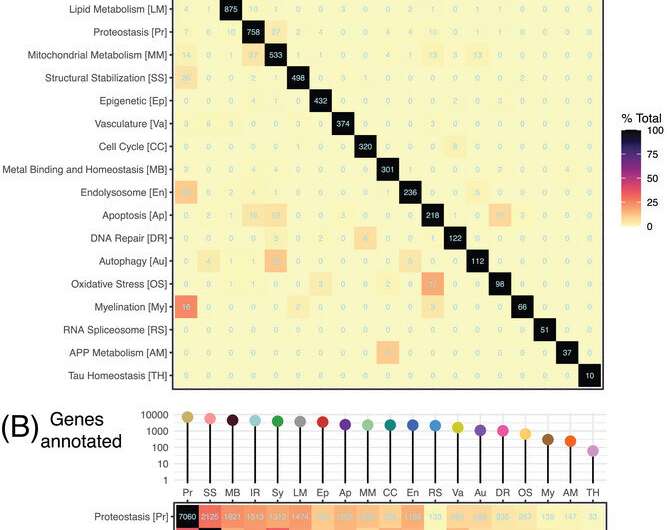
The team used machine learning to draw together and overlay findings from more than two dozen large-scale genetic studies, along with multi-omic analyses of almost 2,900 brains, to identify thousands of potential targets for therapeutic interventions. The targets were then sorted into 19 separate "biodomains" reflecting biological mechanisms believed to contribute to Alzheimer's disease.
Carter and his colleagues didn't just want a long list of undifferentiated gene and protein targets. Instead, each target is associated with a specific therapeutic hypothesis—making it easier to understand how it works, and to identify candidates for experimental validation.
The team was also able to flag targets likely to play a role in the early stages of Alzheimer's, supporting the development of new diagnostic and therapeutic tools for pre-symptomatic interventions.
"This is incredibly important, but also very challenging: most of our data come from post-mortem brains, so our job was like trying to deduce where a forest fire began after everything has been incinerated," said Carter. "Our computer modeling effectively rewinds the progression of the disease to identify early markers that correspond to late-stage pathology."
That approach is already generating important insights, including new evidence that mitochondria—the powerhouse of the cell—could play a significant role in the early stages of Alzheimer's disease. The team found a number of promising targets in this biodomain, suggesting that mitochondrial function could be a very strong early indicator of Alzheimer's—and a key driver of the disease's progression.
The findings and their full dataset are being made publicly available through the Emory-Sage-Structural Genomics Consortium-JAX TREAT-AD Center, part of a consortium dedicated to de-risking Alzheimer's research, giving researchers and biotech innovators a foundational tool to support smarter and more targeted future research.
"We're taking an aggressively open approach," said Carter. "If any biotech or pharmaceutical company wants to pick this up and run with it, they can—and we hope they will."
More information: Gregory A. Cary et al, Genetic and multi‐omic risk assessment of Alzheimer's disease implicates core associated biological domains, Alzheimer's & Dementia: Translational Research & Clinical Interventions (2024). DOI: 10.1002/trc2.12461