Manipulating the microbiome could help manage weight
Vaccines and antibiotics may someday join caloric restriction or bariatric surgery as a way to regulate weight gain, according to a new study focused on the interactions between diet, the bacteria that live in the bowel, and the immune system.
Bacteria in the intestine play a crucial role in digestion. They provide enzymes necessary for the uptake of many nutrients, synthesize certain vitamins and boost absorption of energy from food. Fifty years ago, farmers learned that by tweaking the microbial mix in their livestock with low-dose oral antibiotics, they could accelerate weight gain. More recently, scientists found that mice raised in a germ-free environment, and thus lacking gut microbes, do not put on extra weight, even on a high-fat diet.
In a study, published Aug. 26 in the journal Nature Immunology, a research team based at the University of Chicago was able to unravel some of the mechanisms that regulate this weight gain. They focused on the relationship between the immune system, gut bacteria, digestion and obesity. They showed how weight gain requires not just caloric overload but also a delicate, adjustable—and transmissible—interplay between intestinal microbes and the immune response.
"Diet-induced obesity depends not just on calories ingested but also on the host's microbiome," said the study's senior author Yang-Xin Fu, MD, PhD, professor of pathology at the University of Chicago Medicine. For most people, he said, "host digestion is not completely efficient, but changes in the gut flora can raise or lower digestive efficiency."
So the old adage "you are what you eat" needs to be modified, Fu suggested, to include, "as processed by the microbial community of the distal gut and as regulated by the immune system."
To measure the effects of microbes and immunity, the researchers compared normal mice with mice that have a genetic defect that renders them unable to produce lymphotoxin, a molecule that helps to regulate interactions between the immune system and bacteria in the bowel. Mice lacking lymphotoxin, they found, do not gain extra weight, even after prolonged consumption of a high-fat diet.
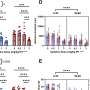
On a standard diet, both groups of mice maintained a steady weight. But after nine weeks on a high-fat diet, the normal mice increased their weight by one-third, most of it fat. Mice lacking lymphotoxin ate just as much, but did not gain weight.
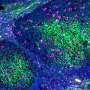
The high-fat diet triggered changes in gut microbes for both groups. The normal mice had a substantial increase in a class of bacteria (Erysopelotrichi) previously associated with obesity and related health problems. Mice that lacked lymphotoxin were unable to clear segmented filamentous bacteria, which has previously been found to induce certain immune responses in the gut.
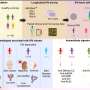
The role of gut microbes was confirmed when the researchers transplanted bowel contents from the study mice to normal mice raised in a germ-free environment—and thus lacking their own microbiome. Mice who received commensal bacteria from donors that made lymphotoxin gained weight rapidly. Those that got the bacteria from mice lacking lymphotoxin gained much less weight for about three weeks, until their own intact immune system began to normalize their bacterial mix.
When housed together, the mice performed their own microbial transplants. Mice are coprophagic; they eat each other's droppings. In this way, the authors note, mice housed together "colonize one another with their own microbial communities." After weeks together, even mice with the immune defect began to gain weight. They also were able to reduce the presence of segmented filamentous bacteria in their stool.
Moving from normal chow to the high-fat diet initiated a series of related changes, the authors found. First, it altered the balance of microbes in the digestive system. These changes in the microbiome altered the immune response, which then introduced further changes to the intestinal microbial community.
These changes "provide inertia for the obese state," the authors said, facilitating more efficient use of scarce food resources.
"Our results suggest that it may be possible to learn how to regulate these microbes in ways that could help prevent diseases associated with obesity," said Vaibhav Upadhyay, first author of the study and an MD/PhD student working in Fu's laboratory. "We now think we could inhibit the negative side effects of obesity by regulating the microbiota and perhaps manipulating the immune response."
Or, 20 years from now, "when there are 10 billion people living on earth and competing for food, we may want to tilt digestive efficiency in the other direction," Fu added.
The authors cautioned, however, that with more than 500 different strains of bacteria present in the gut, "the precise microbes that promote such weight gain and the specific host responses that foster their growth need to be better established."
More information: DOI:10.1038/ni.2403