Study identifies a potential cause of Parkinson's disease

Deciphering what causes the brain cell degeneration of Parkinson's disease has remained a perplexing challenge for scientists. But a team led by scientists from The Scripps Research Institute (TSRI) has pinpointed a key factor controlling damage to brain cells in a mouse model of Parkinson's disease. The discovery could lead to new targets for Parkinson's that may be useful in preventing the actual condition.
The team, led by TSRI neuroscientist Bruno Conti, describes the work in a paper published online ahead of print on November 19, 2012 by the Journal of Immunology.
Parkinson's disease plagues about one percent of people over 60 years old, as well as some younger patients. The disease is characterized by the loss of dopamine-producing neurons primarily in the substantia nigra pars compacta, a region of the brain regulating movements and coordination.
Among the known causes of Parkinson's disease are several genes and some toxins. However, the majority of Parkinson's disease cases remain of unknown origin, leading researchers to believe the disease may result from a combination of genetics and environmental factors.
Neuroinflammation and its mediators have recently been proposed to contribute to neuronal loss in Parkinson's, but how these factors could preferentially damage dopaminergic neurons has remained unclear until now.
Making Connections
Conti and his team were looking for biological pathways that could connect the immune system's inflammatory response to the damage seen in dopaminergic neurons. After searching human genomics databases, the team's attention was caught by a gene encoding a protein known as interleukin-13 receptor alpha 1 chain (IL-13Ra1), as it is located in the PARK12 locus, which has been linked to Parkinson's.
IL-13rα1 is a receptor chain mediating the action of interleukin 13 (IL-13) and interleukin 4 (IL-4), two cytokines investigated for their role as mediators of allergic reactions and for their anti-inflammatory action.
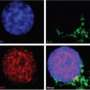
With further study, the researchers made the startling discovery that in the mouse brain, IL-13Ra1 is found only on the surface of dopaminergic neurons. "This was a 'Wow!' moment," said Brad Morrison, then a TSRI postdoctoral fellow and now at University of California, San Diego, who was first author of the paper with Cecilia Marcondes, a neuroimmunologist at TSRI.
Conti agrees: "I thought that these were very interesting coincidences. So we set out to see if we could find any biological significance."
The scientists did—but not in the way they were expecting.
'Something New Going On'
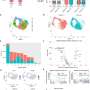
The scientists set up long-term experiments using a mouse model in which chronic peripheral inflammation causes both neuroinflammation and loss of dopaminergic neurons similar to that seen in Parkinson's disease. The team looked at mice having or lacking IL-13Ra1 and then compared the number of dopaminergic neurons in the brain region of interest.
The researchers expected that knocking out the IL-13 receptor would increase inflammation and cause neuronal loss to get even worse. Instead, neurons got better.
"We were very surprised at first," said Conti. "When we stopped to think, we got very excited because we understood that there was something new going on."
Given that cells fared better without the receptor, the team next explored whether damage occurred when dopaminergic neurons that express IL-13Rα1 were exposed to IL-13 or IL-4. But exposure to IL-13 or IL-4 alone did not induce damage.
However, when the scientists exposed the neurons to oxidative compounds, they found that both IL-13 and IL-4 greatly enhanced the cytotoxic effects of oxidative stress.
"This finally helps us understand a basic mechanism of the increased susceptibility and preferential loss of dopaminergic neurons to oxidative stress associated with neuroinflammation," said Marcondes.
The finding also demonstrated that anti-inflammatory cytokines could contribute to neuronal loss. In their article, the authors note they are not suggesting that inflammation is benign but that IL-13 and IL-4 may be harmful to neurons expressing the IL-13Rα1, despite their ability to ultimately reduce inflammation. "One could say that it is not the fall that hurts you, but how you stop," said Conti.
More Clues
Along with these results, additional clues suggest that the IL-13 receptor system could be a major player in Parkinson's. For instance, some studies show Parkinson's as more prevalent in males, and the gene for IL-13Rα1 is located on the X chromosome, where genetic variants are more likely to affect males.
And, though not definitive, other studies have suggested that Parkinson's disease might be more common among allergy sufferers. Since IL-13 plays a role in controlling allergic inflammation, Conti wonders if the IL-13 receptor system might explain this correlation.
If further research confirms the IL-13 receptor acts in a similar way in human dopaminergic neurons as in mice, the discovery could pave the way to addressing the underlying cause of Parkinson's disease. Researchers might, for instance, find that drugs that block IL-13 receptors are useful in preventing loss of dopaminergic cells during neuroinflammation. And, since the IL-13 receptor forms a complex with the IL-4 receptor alpha, this might also be a target of interest. With much exciting research ahead, Conti said, "This is just the beginning."
More information: "IL-13Rα1 expression in dopaminergic neurons contributes to their oxidative stress-mediated loss following chronic systemic treatment with LPS," Journal of Immunology.