Sibling study discovers genetic region linked to control of key blood-clotting protein
In 2006, the lab of Dr. David Ginsburg at the Life Sciences Institute put a call out for siblings attending the University of Michigan to donate blood for a study of blood-clotting disorders.
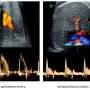
The samples were collected over three years and have now enabled the researchers to identify the specific parts of the genome responsible for levels of a key substance for blood clotting. The findings were reported online Dec. 24 in the Proceedings of the National Academy of Sciences.
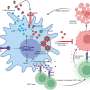
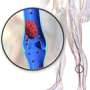
Von Willebrand disease is the most common hereditary blood-clotting disorder—it's more common, but usually milder, than hemophilia. The disease is caused by lower-than-normal levels of von Willebrand factor, a substance that circulates in blood and serves as the "glue" to help blood platelets stick where they're needed to stop bleeding.
While a low level of von Willebrand factor may cause uncontrolled bleeding, high levels can contribute to blood clots, heart attacks and strokes. Age and environmental factors can lead to increased von Willebrand factor, and understanding how the body regulates the substance can help researchers develop treatments for diseases caused by excessive clotting.
Previous studies pinpointed two major genes that partially regulated levels of von Willebrand factor in the blood. However, these two genes only explained a small part of the inherited differences in von Willebrand factor levels between people. Studying siblings in college provided some clues to those differences.
A team of researchers led by Ginsburg, who is a faculty member at the Life Sciences Institute, a professor of internal medicine, human genetics and pediatrics at the U-M Medical School and a Howard Hughes Medical Institute investigator, performed a genetic analysis in two young and healthy cohorts. Because the blood donors were in their early 20s, the effect of other factors known to cause an excess of von Willebrand factor, like age and smoking, were minimized, giving the scientists a better chance of uncovering genetic causes.
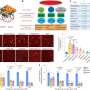
The researchers report that the first part of their analysis, called a genome-wide association study, confirmed the two major genes already known to explain a small part of the differences in von Willebrand factor.
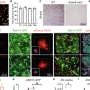
The researchers also looked at which parts of the genome were shared between siblings and how this related to von Willebrand factor levels. They identified a section of chromosome 2 that contains a gene that significantly regulates von Willebrand factor but had not previously been detected in studies of unrelated individuals.
The next step will be to determine the identity of the exact gene on chromosome 2, how it differs among people, and how these differences alter the level of von Willebrand factor and the associated risk for bleeding and blood clotting. Understanding how this gene affects von Willebrand factor may lead to improved diagnosis for bleeding and blood-clotting disorders, as well as new approaches to treatment.
The researchers plan to apply a similar analysis to other traits in blood clotting, says Dr. Karl Desch, assistant professor in the pediatrics department at the U-M Medical School and first author of the study.
"Using the sibling structure in this study, we will hopefully discover more novel loci that determine the variation in these other traits," he said.
More information: www.pnas.org/content/early/201 … 885110.full.pdf+html