This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Gut bacteria enhance cancer immunotherapy in mouse study
Roughly one in five cancer patients benefit from immunotherapy—a treatment that harnesses the immune system to fight cancer. Such an approach to beating cancer has seen significant success in lung cancer and melanoma, among others. Optimistic about its potential, researchers are exploring strategies to improve immunotherapy for cancers that don't respond well to the treatment, with the hope of benefiting more patients.
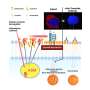
Now, researchers at Washington University School of Medicine in St. Louis have found, in mice, that a strain of gut bacteria—Ruminococcus gnavus—can enhance the effects of cancer immunotherapy. The study, which appears May 17 in Science Immunology, suggests a new strategy of using gut microbes to help unlock immunotherapy's untapped cancer-fighting potential.
"The microbiome plays an important role in mobilizing the body's immune system to attack cancer cells," explained the study's senior author, Marco Colonna, MD, the Robert Rock Belliveau, MD, Professor of Pathology.
"Our findings shine a light on one bacterial species in the intestine that helps an immunotherapy drug eliminate tumors in mice. Identifying such microbial partners is an important step in developing probiotics to help improve the effectiveness of immunotherapy drugs and benefit more cancer patients."
Cancer immunotherapy employs the body's immune cells to target and destroy tumors. One such treatment uses immune checkpoint inhibitor drugs to unleash the immune system by releasing the natural brakes that keep immune T cells quiet, a feature that prevents the body from harming itself. But some tumors fight back to suppress the attacking immune cells, damping the effectiveness of such inhibitors.
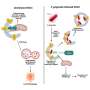
Colonna and first co-author Martina Molgora, Ph.D., a postdoctoral researcher, previously established a collaboration with colleague Robert D. Schreiber, Ph.D., the Andrew M. and Jane M. Bursky Distinguished Professor, in which they completely eliminated sarcoma tumors in mice using a two-pronged inhibition approach.
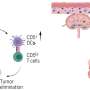
The researchers inhibited TREM2, a protein made by tumor macrophages to stop T cells from attacking the growing tumor. They then showed that a cancer immunotherapy drug was more effective when TREM2 was blocked. The result indicated that TREM2 dampens immunotherapy's efficacy.
In an experiment that formed the basis of the new study, the researchers made a surprise observation. TREM2 mice had the same beneficial response to the checkpoint inhibitor when they were housed with mice lacking the protein. This result came about when the researchers deviated from their typical protocol of separating the mice before treating them with the inhibitor.
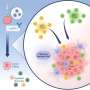
Cohabiting mice share microbes with one another. The researchers suspected the effects might have been due to exchanges of gut bacteria.
The researchers worked with Jeffrey I. Gordon, MD, the Dr. Robert J. Glaser Distinguished University Professor, and co-first author Blanda Di Luccia, Ph.D., a postdoctoral researcher, to study the microbes in the intestines of the mice that were treated successfully with immunotherapy. They found an expansion of Ruminococcus gnavus, compared with a lack of such microbes in mice that didn't respond to the therapy.
R. gnavus has been found in gut microbiota of cancer patients who respond well to immunotherapy, Colonna explained. In clinical trials, fecal transplants from such individuals have helped some unresponsive patients reap immunotherapy's benefits.
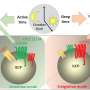
The researchers, including co-first author and graduate student Darya Khantakova, introduced R. gnavus to the mice and then treated the tumors with a checkpoint inhibitor. The tumors shrank, even when TREM2 was available as a weapon to dampen the effect of the immunotherapy.
Gordon, director of the Edison Family Center for Genome Sciences & Systems Biology, noted that evidence is mounting that the microbiota boosts immunotherapy. Identifying relevant species, such as R. gnavus, could lead to a next-generation probiotic that could synergize with immunotherapy to improve cancer care, he explained.
The scientists next aim to understand how R. gnavus assists in tumor rejection, which may reveal new ways to help cancer patients. For example, if the microbe produces an immune-activating metabolite through the process of digesting food, that knowledge opens the opportunity to use metabolites as immunotherapy enhancers.
Microbes also can leak out of the gut and trigger an immune response in the tumor or activate gut T cells that migrate to the tumor to mount an attack, Colonna explained. The researchers are exploring the three possibilities.
More information: Blanda Di Luccia et al, TREM2 deficiency reprograms intestinal macrophages and microbiota to enhance anti-PD-1 tumor immunotherapy, Science Immunology (2024). DOI: 10.1126/sciimmunol.adi5374. www.science.org/doi/10.1126/sciimmunol.adi5374