Fasting time for tumour cells
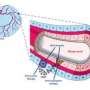
(Medical Xpress)—Tumours need a steady supply of sufficient nutrients to be able to grow. In order to secure the nutrient availability, they secrete messenger compounds to stimulate neighbouring blood vessels to proliferate and sprout. Scientists from the Max Planck Institute for Neurological Research in Cologne, Germany, have now identified a new positive feedback loop involving the Vascular Endothelial Growth Factor (VEGF) and its receptor 'VEGFR-2' in human lung adenocarcinoma.
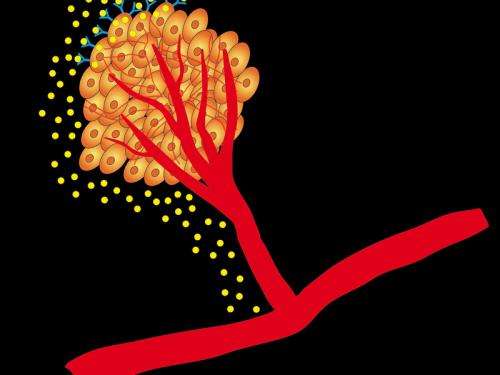
When VEGF binds to VEGFR-2 on cancer cells, secretion of the growth-factor itself is boosted consequently accelerating tumour growth. In experiments on mice with lung cancer, the scientists switched off the growth-factor and proteins responsible for this signalling thereby slowing down tumour growth. The tumours were even reduced in size by employing other inhibitors in combination. The researchers thus proved the existence of VEGF Receptor-2 on tumour cells and described a new signalling pathway in lung cancer cells that triggers the sprouting of new blood vessels. In addition, they also learnt from examinations of lung cancer patients that therapy with these inhibitors only makes sense if the cancer cells express large numbers of VEGFR2. These results can contribute to developing new cancer therapies.
Tumour cells often remain in a dormant state before dividing in an uncontrolled manner. During this dormant period, approximately as many cells die off as regenerate. Change in the tumour cell genetics leads to messenger compounds being secreted that stimulate proliferation of blood vessels. Only then the tumour can begin to expand. Without this transition from dormant to active state, the growth of cancer cells would be limited to a dimension harmless for the body.
Scientists at the Max Planck Institute for Neurological Research want to use this as a point of attack for tumour therapy. To this end, they investigated the effect of VEGF, which enables blood vessels to expand. According to the research group led by Roland Ullrich, VEGF also acts directly on the secreting tumour cells. These cells re-absorb it via VEGFR-2 and thus produce even more VEGF. "This positive feedback loop causes more and more new blood vessels to generate and the cancer to grow even faster", explains Ullrich. "We therefore wanted to find out what happens when we interrupt them." The scientist's idea: "to cut off the tumour from the supply of nutrients – simply starving it out". Ullrich compares the fight against cancer with the siege of a fortress: "You don't have to necessarily storm the castle to overcome the enemy. It is sufficient to turn off the water-supply."
In the second part of their study, they therefore experimented with mice that exhibited a specific form of lung cancer and blocked their VEGFR-2. "And we did detect a deceleration of the cancer growth in the animals", says Ullrich. "An even more impressive result was obtained when we administered an additional inhibitor." This inhibitor disrupts what is known as the MAPK signalling pathway, which drives growth of tumour cells, among other things. Administered individually, both inhibitors only slowed down the cancer growth, while in combination they were able to actually shrink the tumour. The scientists therefore assume a connection between the MAPK signalling pathway and VEGFR2 inhibition. "If the supply of nutrients to the cells is cut off, they simply begin to grow more vigorously", explains Ullrich, "and we can prevent that with the help of the second inhibitor."
However, not all types of cancer are suited to this kind of treatment. These inhibitors can only be effective if the cancer cells also express the receptors for VEGF (VEGFR-2) on their membranes in moderate to high numbers. According to the results of the Cologne scientists, about every fifth lung cancer patient has VEGFR-2 on the tumour cells and could therefore be treated in this way.
More information: Chatterjee, S. et al. Tumor VEGF: VEGFR2 autocrine feed-forward loop triggers angiogenesis in lung cancer, J Clin Invest. 2013;123(4):xxxx–yyyy. doi:10.1172/JCI65385