July 10, 2013 report
Exosomal transmission of viral resistance in Hepatitis B
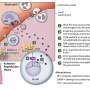
(Medical Xpress)—To move material in bulk, the standard shipping container used by cells, is the vesicle. These approximately 40-micron sized spheres are essentially recyclable grocery bags that can be loaded and adorned with a huge variety of molecules. Exosomes are a particular kind of vesicle that get released when structures known as multivesicular bodies (MSVs) fuse with the plasma membrane. They can potentially be used for diagnostics and perhaps as treatment for many kinds of disease. A paper published in Nature Immunology, has recently shown that exosomes may have a natural function acting against the Hepatitis B virus (HBV). The Chinese authors on the paper have demonstrated that cell-to-cell transmission of the natural antiviral agent, type 1 Interferon (IFN), is mediated by exosomes.
Exosomes have been shown to serve as mediators of intercellular communication and are capable of horizontally transferring mRNA and microRNA. The authors hypothesized that exosomal transfer could explain interferon-induced antiviral responses despite the documented inhibition of interferon-stimulated gene expression by the virus. That is quite a delicate theory applied to a complex system with many diverse players.
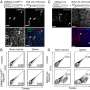
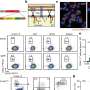
IFN, at least the alpha subtype, is given for treatment of chronic HBV infection, but it does not appear to directly inhibit HBV replication. The liver's immune system is composed of natural killer cells, T cells, and its own resident macrophages, the Kupffer cells. The authors used a cell-isolated culture system to determine whether the IFN antiviral response could be transmitted from these various types of nonparenchymal liver cells to the liver's hepatocytes. In addition, they were able to show that in-vivo exosomal transfer of antiviral molecules against HBV also occurs.
Blocking exosomes increased hepatitis in mice, suggesting perhaps, that augmenting exosome release could be beneficial in treating the disease. Exosomes have been shown to contain a huge variety of anti-viral molecules, which makes it difficult for the virus to deal with all of them together. The authors used a "knockdown and rescue" strategy to further explore the role of nonparenchymal liver cells in the antiviral effects of IFN. In particular, they targeted the STAT1 transcriptional activator with a vector encoding short hairpin RNA. The expression products of interferon-stimulated genes under the control of interferon-stimulated response elements were shown to establish an antiviral state in target cells.
The researchers also collected IFN stimulated and unstimulated exosomes by centrifugation and analyzed their components. They used microarray identification of mRNA and microRNA to reveal their differential expression. They hypothesized that the changes reflect preferential sorting of specific exosomal cargo.
The mechanisms by which exosomes are loaded with the proper molecules, when needed, are largely unknown, as is their targeting and timing of release. Neurons have made an art of vesicle communication and perhaps some clues will be found from the study of these, and other, systems. The authors note that the relative importance of different antiviral pathways involving IFN likely depends on the specifics of the virus-host interaction. In addition to the roles of the Kupffer cells demonstrated here, many other cells types are likely to be involved as well.
The manner in which exosomal molecules inhibit HBV is also largely a mystery. It is possible however, that the exosomal method of attack is important in other diseases as well. One study, for example, has reported that APOBEC3G can be packaged and delivered via exosomes to restrict the replication of HIV. Inhibiting exosome release has also been shown to impair the effect of IFN against the adenovirus. The many other dangerous viruses against which we currently have little protection suggests that the study of these new methods of dealing with them may be a worthwhile pursuit.
More information: Exosomes mediate the cell-to-cell transmission of IFN-?-induced antiviral activity, Nature Immunology (2013) doi:10.1038/ni.2647
Abstract
The cell-to-cell transmission of viral resistance is a potential mechanism for amplifying the interferon-induced antiviral response. In this study, we report that interferon-? (IFN-?) induced the transfer of resistance to hepatitis B virus (HBV) from nonpermissive liver nonparenchymal cells (LNPCs) to permissive hepatocytes via exosomes. Exosomes from IFN-?-treated LNPCs were rich in molecules with antiviral activity. Moreover, exosomes from LNPCs were internalized by hepatocytes, which mediated the intercellular transfer of antiviral molecules. Finally, we found that exosomes also contributed to the antiviral response of IFN-? to mouse hepatitis virus A59 and adenovirus in mice. Thus, we propose an antiviral mechanism of IFN-? activity that involves the induction and intercellular transfer of antiviral molecules via exosomes.
© 2013 Medical Xpress