Pitt team aims to change tissue microenvironment to fend off cancerous tumors
The magic to killing cancer cells might not be in the bullet, but in the gunpowder that accompanies it, according to researchers at the University of Pittsburgh Cancer Institute (UPCI), partner with UPMC CancerCenter. Instead of developing new drugs that directly target tumors, they have been working on adjuvant agents that alter the immunological microenvironment around the tumor to boost the effectiveness of existing drugs and the effectiveness of each patient's immune responses against cancer.
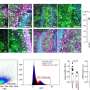
While multiple studies worldwide have demonstrated that the infiltration of tumor tissues with immune killer cells called cytotoxic T cells (CTLs) improves the prognosis of cancer patients, it remains unclear how to manipulate tumor microenvironment to promote CTL infiltration.
In a recent presentation at the Cancer Vaccines and Gene Therapy Meeting in Malvern, Pa., Pawel Kalinski, M.D., Ph.D., professor of surgery, University of Pittsburgh School of Medicine, presented laboratory data in support of the clinical use of "combinatorial adjuvants" that induce desirable patterns of tumor-associated inflammation and result in CTL infiltration into tumors. He also discussed preliminary data from a phase I/II trial led by Amer Zureikat, M.D., an assistant professor of surgery at Pitt, in which colorectal cancer patients were given escalating doses of interferon alpha (IFN-α) in combination with a COX2 blocker and Ampligen, an experimental toll-like receptor-3 (TLR3) ligand, factors that synergistically enhance IFN-α's immunological effects in tumor microenvironments.
"The first part of the trial is not complete, but so far it appears that the treatment was well-tolerated," Dr. Kalinski said. "Our early observations are completely consistent with our preclinical predictions and hint that the combination therapy may be altering the tumor environment to make it more susceptible to immune attack. We are hopeful that the next, randomized part of the study will directly demonstrate the difference between the tumors of patients receiving standard treatment of chemotherapy and surgery and the patients receiving additional immunotherapy, which would lead us to expect a therapeutic benefit to this strategy."
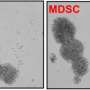
Tumors are typically able to undermine the body's immune defenses, sending out cellular signals that call in regulatory T-cells to suppress the activity of natural killer cells, he explained. While there are many adjuvants available to enhance immune system response, most are nonselective and, therefore, ineffective. Preclinical experiments conducted by Dr. Kalinski's team indicate that the combination of IFN-α, a COX2 inhibitor and TLR3 ligands selectively brings the right kinds of immune cells to the tumor to produce what he calls "good inflammation." Such a treatment preferentially induces good inflammation within tumors, allowing the immune system to selectively attack cancer cells without inadvertently harming healthy tissues nearby.
In the second part of the study, which could begin at the beginning of next year, colorectal cancer patients will be randomly assigned to receive either conventional treatment with chemotherapy followed by surgical removal of the tumor, or to additionally receive two cycles of the IFN-α-based chemokine-modulatory regimen between chemo and surgery.
"After surgery, we will be able to compare the tumors from the two groups to see if there is a difference in their immunological microenvironment that could be beneficial," Dr. Kalinski said. "This adjuvant strategy might also work for many other kinds of cancers because it's not targeting specific tumor markers, but boosting the immune system's own ability to find cancer cells and destroy them."
He added that future efforts would aim to add a vaccine component based on dendritic cells, which recognize foreign proteins to stimulate an immune response against them. That strategy, too, would rely on boosting the body's own defenses rather than providing a specific tumor marker as a target.