Common mutation is culprit in acute leukemia relapse
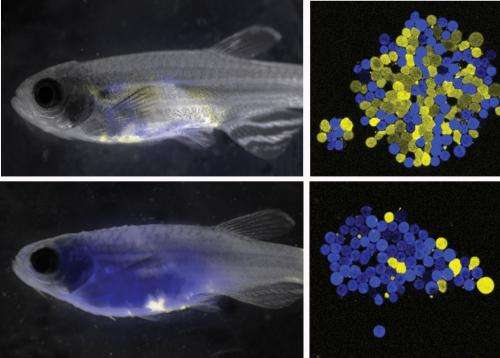
Harvard stem cell scientists have identified a mutation in human cases of acute lymphoblastic leukemia that likely drives relapse. The research, published in Cancer Cell, could translate into improved patient care strategies for this particular blood cancer, which typically affects children but is more deadly in adults.
In recent years, a trend toward single-cell analysis has shown that individual cells within a tumor are capable of amassing mutations to make them more aggressive and treatment resistant. So while 99% of a tumor may be destroyed by the initial treatment, a particularly aggressive cell can survive and then cause a cancer patient with the "all clear" to relapse six months later.
Harvard Stem Cell Institute Principal Faculty member David Langenau, PhD, and his lab members in the Department of Pathology at Massachusetts General Hospital used zebrafish to search for these rare, relapse-driving leukemia cells and then designed therapies that could kill these cells.
The researchers found that at least half of relapse-driving leukemic cells had a mutation that activated the Akt pathway, which rendered cells resistant to common chemotherapy and increased growth. From that insight, Langenau's lab next examined human acute lymphoblastic leukemia and discovered that inhibition of the Akt pathway restored leukemic cell responses to front-line chemotherapy.
"The Akt pathway appears to be a major driver of treatment resistance," Langenau said. "We also show that this same pathway increases overall growth of leukemic cells and increases the fraction of cells capable of driving relapse."
Jessica Blackburn, PhD, the study's first author adds, "Our work will likely help in identifying patients that are prone to relapse and would benefit from co-treatment with inhibitors of the Akt pathway and typical front-line cancer therapy."
In addition to determining how best to translate this finding into the clinic, Langenau hopes to to identify other mutations that lead to relapse. The work should identify a host of other potential drug targets for patients with aggressive leukemia.
The research took five-and-a-half years to complete, and has its origins as one of the first projects Langenau took on when he started his laboratory. The study was also the most labor-intensive project his lab members took on, with over 6,000 zebrafish transplant experiments.
More information: Clonal evolution enhances leukemia-propagating cell frequency in T-cell acute lymphoblastic leukemia through Akt/mTORC1 pathway activation. Cancer Cell. March 6, 2014















