Unravelling nerve-cell death in rare children's disease
A team of scientists, led by Stuart Lipton, M.D., Ph.D., professor and director of the Neuroscience and Aging Research Center at Sanford-Burnham Medical Research Institute (Sanford-Burnham), recently discovered why cerebellar granule cell neurons in patients suffering from ataxia-telangiectasia (A-T) were unable to repair DNA damage and thus died.
A-T is a hereditary condition that begins early in childhood, and causes a gradual loss of certain nerve cells in the cerebellum of the brain. A-T occurs in about 1 in 40,000 births, with symptoms including severe loss of muscle control, dilated small blood vessels, repeated infections in the sinus and lungs, and it frequently leads to cancers such as lymphoma or leukemia. Today, thanks to improved treatment of infections and cancer, many patients live into their 30s or longer.
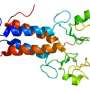
His study, with Nobuki Nakanishi, Ph.D., associate professor in Sanford-Burnham's Degenerative Disease Program, was published March 25 in The Journal of Neuroscience. The researchers used genetically engineered mice to show that myoctye-enhancer factor 2D (MEF2D), a transcription factor that turns on specific genes involved in cell survival, is activated after binding to a protein known as A-T mutated (ATM). When the ATM gene that codes for the ATM protein is mutated, thus causing A-T, ATM-MEF2D-survival signals in response to DNA damage are ineffective and may contribute to neurodegeneration.
"This is the first time that a signal that regulates MEF2D-dependent survival in response to DNA damage has been identified," said Lipton. "Knowing that ATM-mediated activation of MEF2D promotes survival in cerebellar neurons in response to DNA damage may provide a therapeutic opportunity for A-T. For example, if we can confirm that defects in the ATM-MEF2D signal contribute to A-T, we can search for molecules that enhance MEF2D activity to 'revive' the DNA repair system."
"As parents, we are excited that this research could lead to new ideas about how to slow the brain cell loss seen in our kids, improving their ability to walk, talk, and eat. This could lead to big improvements in their quality of life," said Brad Margus, voluntary president and founder of the A-T Children's Project.
In general, DNA repair systems are essential for cellular integrity and stability. DNA can be damaged in many ways, including UV rays, tobacco, and oxidative damage from byproducts of metabolism, such as free radicals. Inherited defects of DNA repair systems can lead to many types of cancer, dwarfism, growth and mental retardation, deafness—as well as A-T.
"Prior to this study, we knew that p53—a protein known as the guardian angel of the genome—was a target of ATM activation and contributes to the control and efficacy of DNA repair. Now, we have shown another pathway whereby MEF2D participates in DNA damage repair in the cerebellum. The fact that there is an abundance of MEF2D in the cerebellum, and that ATM mutations are associated with A-T, adds support to the proposed ATM-MEF2D dysfunction as a cause of A-T," said Lipton.
"Moving forward, we will be interested to see if this mechanism contributes to other rare hereditary diseases with defects in DNA repair."