March 18, 2014 report
Research shows breast cancer gene affects brain development
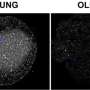
(Medical Xpress)—The BRCA1 gene, known for its role in suppressing the growth of breast and ovarian tumors, could be necessary for brain development. In a study appearing in the Proceedings of the National Academy of Sciences, Inder Verma and his colleagues at the Salk Institute for Biological Studies in La Jolla, California created a strain of mice lacking BRCA1 in neuronal stem cells. These mice had smaller than normal brains with numerous defects. The researchers think BRCA1 played an important role in the evolution of the mammalian brain.
BRCA1 helps repair DNA damage in dividing cells. Women with mutated copies of this gene have a high risk of developing ovarian and breast cancer. However, BRCA1 is most highly expressed in the embryonic neuroepithelium, cells in the embryo that will later form the nervous system.
The researchers hypothesized that the high level of BRCA1 activity in neuronal stem cells means that BRCA1 is essential for healthy brain development. Embryonic and adult neuronal stem cells are very susceptible to damage from ionizing radiation. Verma's team thought the BRCA1 gene might help prevent this damage.
To test their hypothesis, the team created a strain of genetically modified mice whose neuronal stem cells did not have any BRCA1. After the mice were born, they had problems with muscle coordination and became unusually agitated when separated from their mothers. When the researchers examined the brains of these mice, they found defects in the neocortex, hippocampus and cerebellum, structures involved in cognition and motor learning, as well as in the olfactory bulb. All of these structures were much smaller than normal. The cerebellum was reduced to less than half the normal size.
The researchers found that BRCA1 prevents the protein ATM kinase from destroying dividing cells. ATM kinase kills off cells with double strand DNA breaks unless BRCA1 suppresses its activity. This explains why the brains of these mice experienced unusually high levels of cell death. When the team created another strain of mice whose neuronal stem cells had neither BRCA1 nor ATM kinase, they found these mice appeared no different from control mice.
Verma and his team think BRCA1 was involved in the evolution of mammalian brain size. People with primary microcephaly suffer from a mutation in the gene MCPH1, which regulates levels of BRCA1. These people have brains about the same size as the brains of chimpanzee and bonobos.
More information: Role of BRCA1 in brain development, PNAS, Gerald M. Pao, DOI: 10.1073/pnas.1400783111
Abstract
Breast cancer susceptibility gene 1 (BRCA1) is a breast and ovarian cancer tumor suppressor whose loss leads to DNA damage and defective centrosome functions. Despite its tumor suppression functions, BRCA1 is most highly expressed in the embryonic neuroepithelium when the neural progenitors are highly proliferative. To determine its functional significance, we deleted BRCA1 in the developing brain using a neural progenitor–specific driver. The phenotype is characterized by severe agenesis of multiple laminated cerebral structures affecting most notably the neocortex, hippocampus, cerebellum, and olfactory bulbs. Major phenotypes are caused by excess apoptosis, as these could be significantly suppressed by the concomitant deletion of p53. Certain phenotypes attributable to centrosomal and cell polarity functions could not be rescued by p53 deletion. A double KO with the DNA damage sensor kinase ATM was able to rescue BRCA1 loss to a greater extent than p53. Our results suggest distinct apoptotic and centrosomal functions of BRCA1 in neural progenitors, with important implications to understand the sensitivity of the embryonic brain to DNA damage, as well as the developmental regulation of brain size.
© 2014 Medical Xpress