A more efficient production process for influenza vaccines could help to cope with future pandemics
Influenza pandemics, such as the 2009 H1N1 'swine flu' outbreak, pose a serious risk to the global population. Vaccination is one route to protection but current manufacturing methods for vaccines limit the volume and speed of production. Now, an international team of researchers including A*STAR's Program in Translational Research on Infectious Disease, Experimental Therapeutics Centre and Singapore Immunology Network, has developed a more efficient production process.
In 2009, it took five months for the vaccine against the H1N1 virus to become available, and the number of doses made was sufficient for only a fraction of the global population. Both shortfalls were down to the conventional vaccine production process and so the research team wanted to develop a more efficient technique.
"The licensed H1N1 flu vaccine is made by growing the influenza virus in chicken eggs," explains David Skibinski, the study's lead author. In comparison, "our vaccine is produced by expressing the main surface antigen of influenza in bacteria."
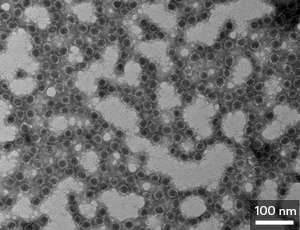
The researchers attached the antigen—part of a protein from the surface of the H1N1 virus—to harmless virus-like particles and injected them into mice. Compared with the mice that were inoculated with the licensed vaccine, animals inoculated with the new vaccine produced a similar number of effective antibodies against the H1N1 virus, which demonstrated that the new production method provided immunity as effectively as the existing vaccine.
There was, however, an extra advantage: mice that were inoculated with the new vaccine produced more T cells than mice that were inoculated with the licensed vaccine. T cells play an important role in protecting the body against influenza, reduce the severity of the disease, and provide protection against different strains of the virus.
Skibinski says the new way of producing the influenza vaccine could improve the global response to future pandemics by shortening the time between the emergence of a virus and production of a functional vaccine, thus enabling individuals to be vaccinated before they become infected. "Greater cost-efficiency and yields would also enable many smaller or developing nations to manufacture their own vaccine," adds Skibinski.
Having shown the new vaccine's effectiveness in mice, the research team's next step was to see if the findings could be transferred to humans. So far, the results are positive, reports Skibinski. "A clinical trial in humans under the direction of the A*STAR D3 unit has demonstrated that the vaccine is safe, well tolerated and induces antibody responses comparable to those of approved seasonal influenza vaccines."
More information: Skibinski, D. A. G., Hanson, B. J., Lin, Y., von Messling, V., Jegerlehner, A. et al. "Enhanced neutralizing antibody titers and Th1 polarization from a novel Escherichia coli derived pandemic influenza vaccine." PLoS ONE 8, e76571 (2013). dx.doi.org/10.1371/journal.pone.0076571