Zika infection may affect adult brain cells
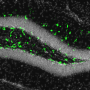
Concerns over the Zika virus have focused on pregnant women due to mounting evidence that it causes brain abnormalities in developing fetuses. However, new research in mice from scientists at The Rockefeller University and ...
Aug 18, 2016
5
3021