RNA-associated introns guide nerve-cell channel production
Researchers at the University of Pennsylvania School of Medicine have discovered that introns, or junk DNA to some, associated with RNA are an important molecular guide to making nerve-cell electrical channels. Senior author James Eberwine, PhD, Elmer Bobst Professor of Pharmacology, and lead authors Kevin Miyashiro, and Thomas J. Bell, PhD, both in Eberwine’s lab, report their findings in this week's early online edition of the Proceedings of the National Academy of Sciences.
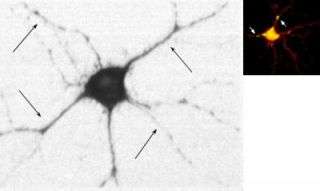
In nerve cells, some ion channels are located in the dendrite, which branch from the cell body of the neuron. Dendrites detect the electrical and chemical signals transmitted to the neuron by the axons of other neurons. Abnormalities in the dendrite electrical channel are involved in epilepsy, neurodegenerative diseases, and cognitive disorders, among others.
Introns are commonly looked on as sequences of "junk" DNA found in the middle of gene sequences, which after being made in RNA are simply excised in the nucleus before the messenger RNA is transported to the cytoplasm and translated into a protein. In 2005, the Penn group first found that dendrites have the capacity to splice messenger RNA, a process once believed to only take place in the nucleus of cells.
Now, in the current study, the group has found that an RNA encoding for a nerve-cell electrical channel, called the BK channel, contains an intron that is present outside the nucleus. This intron plays an important role in ensuring that functional BK channels are made in the appropriate place in the cell.
When this intron-containing RNA was knocked out, leaving the maturely spliced RNA in the cell, the electrical properties of the cell became abnormal. “We think the intron-containing mRNA is targeted to the dendrite where it is spliced into the channel protein and inserted locally into the region of the dendrite called the dendritic spine. The dendritic spine is where a majority of axons from other cells touch a particular neuron to facilitate neuronal communication” says Eberwine. “This is the first evidence that an intron-containing RNA outside of the nucleus serves a critical cellular function.”
“The intron acts like a guide or gatekeeper,” says Eberwine. “It keys the messenger RNA to the dendrite for local control of gene expression and final removal of the intron before the channel protein is made. Just because the intron is not in the final channel protein doesn’t mean that it doesn’t have an important purpose.”
The group surmises that the intron may control how many mRNAs are brought to the dendrite and translated into functional channel proteins. The correct number of channels is just as important for electrical impulses as having a properly formed channel.
The investigators believe that this is a general mechanism for the regulation of cytoplasmic RNAs in neurons. Given the central role of dendrites in various physiological functions they hope to relate this new knowledge to understanding the molecular underpinnings of memory and learning, as well as components of cognitive dysfunction resulting from neurological disease.
Source: University of Pennsylvania