Cell death unleashes full force of human antiviral system
A scientific team led by researchers at the University of Geneva (UNIGE) and the Charite Berlin Medical University has made a completely unprecedented discovery showing how much our immune system is provoked into action when confronted by viral intruders. The possibility of exploiting this mechanism in vaccines holds promise for the development of new ways of preventing and treating infectious diseases and cancer. The discovery is described in today's edition of the journal Science.
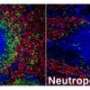
Killer T cells – also called cytotoxic T lymphocytes – play a central role in our immune system. They are capable of specifically identifying and killing cells harbouring viruses or bacteria and also cancer cells. It is likely that cytotoxic T lymphocytes will play an important role in the future development of vaccines against HIV/AIDS, hepatitis C, malaria and also cancer.
It has long been known that viral infection in humans is followed by formation of an army of T lymphocytes. But how does this mobilization come about and who decides it? Researchers think that the alert is provided by dendritic cells capable of identifying a virus by its specific, characteristic molecular motifs called pathogen-associated molecular patterns or PAMPs. After identifying a pathogen, the dendritic cells then coordinate the action of cytotoxic T lymphocytes.
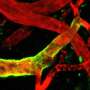
However, another fundamental mechanism underlies the counter-attack launched by the immune system. This mechanism is described for the first time by Professor Max Loehning (Charité Berlin Medical University) and Professor Daniel Pinschewer (UNIGE) in an article published today in the journal Science. It is engaged after the virus has already killed certain cells. These cells release all kinds of molecules when they die, including some very distinctive molecules that experts have dubbed damage-associated molecular patterns or DAMPs. Among the molecular debris of this miniature battlefield, DAMPs are unique in providing an unequivocal signal that the body has suffered damage.
The research team suspects that the ferocity of the attack mounted by the cytotoxic T lymphocytes is strongly related to the presence of a particular DAMP called interleukin 33 (IL-33). This protein is normally found inside cells that line the walls of the spleen and lymph nodes, the "homes" of cytotoxic T lymphocytes inside the body. Therefore, when these cells are killed by viruses, the interleukin 33 they release immediately signals the cytotoxic T lymphocytes to launch an all-out attack.
The authors were able to demonstrate the existence of this mechanism with the aid of mice whose immune system is not able, for genetic reasons, to identify interleukin 33. As expected, these animals are clearly less able to fend off a viral infection. The cytotoxic T lymphocytes struggle to mount an effective counter-attack because there are simply not enough of them.
If the inability of an organism to recognize interleukin 33 makes it more vulnerable to infections of any kind, one could imagine that, conversely, interleukin 33 could be used to increase the number and the aggressiveness of T lymphocytes mobilized during vaccination.
As explained by Max Loehning and Daniel Pinschewer, the alerts provided by PAMPs and DAMPs seem to be complementary during the preparation of a counter-attack: "PAMPs alert dendritic cells which, in turn, enlist killer T cells. However, the response of the latter cells remains fairly mild until they are roused into action by the presence of DAMP released by victims of the virus in the immediate vicinity."
These results represent fertile new territory for researchers developing vaccines against infectious diseases and cancer.