New endoscope technology paves the way for 'molecular-guided surgery' for cancer
(PhysOrg.com) -- With more than 15 million endoscope procedures done on patients each year in the U.S. alone, scientists today reported evidence that a new version of these flexible instruments for diagnosing and treating disease shows promise for helping surgeons more completely remove cancerous tumors. Their report on technology that combines the endoscope with the phenomenon responsible for the eerie blue glow in the cooling water of nuclear reactors was part of the 243rd National Meeting & Exposition of the American Chemical Society (ACS).
“The advance marries endoscopes to one of the newest and most exciting fields of medical imaging,” said Zhen Cheng, Ph.D., who led the research. “Endoscopes are medical devices consisting of a long, thin, flexible tube of optical fibers fitted with a light and a video camera. Doctors can insert them through natural openings in the body or small surgical incisions to diagnose and treat a wide range of diseases. The new approach expands the use of those fibers to include molecular-guided surgery to remove more of a cancerous tumor than is currently possible.”
Conventional endoscopic methods allow doctors to see inside the stomach or colon, for instance, and take biopsy samples for tests. The new imaging technology — called Cerenkov Luminescence Endoscopy (CLE) — has advantages over both traditional endoscopic and imaging techniques, like MRI, in also providing information about the functioning of the tissue, Cheng added.
Cheng, who is with Stanford University, explained that CLE relies on the same phenomenon responsible for the soft blue glow in the cooling water in the core of nuclear power reactors. It results from interactions that occur when invisible particles from the nuclear reaction in the core zip through the water faster than the speed of light in water. The glow was named for its discoverer, a Russian scientist, who won the 1958 Nobel Prize.
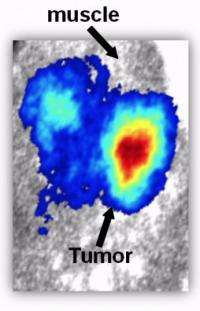
Cerenkov Luminescence Imaging (CLI) emerged just a few years ago when scientists discovered ways to harness the effect in devices that do not involve nuclear power reactors. Excitement grew when laboratory experiments established that CLI could produce images of organs and guide surgery in laboratory animals to remove remaining cancer cells that otherwise would have been invisible to surgeons. Scientists also found that CLI could dramatically improve the resolution of PET scans, enabling PET scanners to detect smaller objects than previously possible. Cerenkov luminescence was especially exciting because the light used to reveal diseased tissue is visible light that can be detected with simple optical sensors. It also is compatible with commercially available optical imaging instruments and a wide selection of the nuclear imaging agents that doctors use to make structures in the body visible.
“One drawback however, is that the weak blue light — unlike the X-rays in other medical scans — barely penetrates through deep tissues,” Cheng said. “This limits the usefulness of the technology in humans, where many tumors develop in areas deep inside the body. Our marriage of Cerenkov luminescence with the endoscope may be the perfect solution. With endoscopy, we can get close enough to the diseased tissue to take advantage of this technology.”
Cheng pointed out that CLE’s transition from laboratory to operating room, which may occur during the next few years, should be relatively smooth because of its similarity to traditional endoscopy. “Since the CLE system is an upgraded version of endoscopy, it is highly compatible with conventional endoscopic imaging requirements,” he said.
The scientists acknowledged funding from the National Institutes of Health, the Department of Defense, the Canary Foundation, the Center for Biomedical Imaging at Stanford and the Friends for an Earlier Breast Cancer Test.