Easy, effective therapy to restore sight: Engineered virus will improve gene therapy for blinding eye diseases
Researchers at the University of California, Berkeley, have developed an easier and more effective method for inserting genes into eye cells that could greatly expand gene therapy to help restore sight to patients with blinding diseases ranging from inherited defects like retinitis pigmentosa to degenerative illnesses of old age, such as macular degeneration.
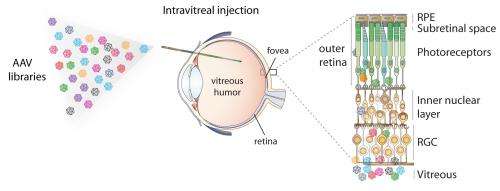
Unlike current treatments, the new procedure – which takes a little as 15-minutes – is surgically non-invasive, and it delivers normal genes to difficult-to-reach cells throughout the entire retina.
Over the last six years, several groups have successfully treated people with a rare inherited eye disease by injecting a virus with a normal gene directly into the retina of an eye with a defective gene. Despite the invasive process, the virus with the normal gene was not capable of reaching all the retinal cells that need fixing.
"Sticking a needle through the retina and injecting the engineered virus behind the retina is a risky surgical procedure," said David Schaffer, professor of chemical and biomolecular engineering and director of the Berkeley Stem Cell Center at the University of California, Berkeley. "But doctors have no choice because none of the gene delivery viruses can travel all the way through the back of the eye to reach the photoreceptors – the light sensitive cells that need the therapeutic gene."
"Building upon 14 years of research, we have now created a virus that you just inject into the liquid vitreous humor inside the eye and it delivers genes to a very difficult-to-reach population of delicate cells in a way that is surgically non-invasive and safe. It's a 15-minute procedure, and you can likely go home that day."
The engineered virus works far better than current therapies in rodent models of two human degenerative eye diseases, and can penetrate photoreceptor cells in the eyes of monkeys, which are like those of humans.
Schaffer and his team are now collaborating with physicians to identify the patients most likely to benefit from this gene-delivery technique and, after some preclinical development, hope soon to head into clinical trials, he said.
Schaffer and John Flannery, UC Berkeley professor of molecular and cell biology and of optometry, along with colleagues from UC Berkeley's Helen Wills Neuroscience Institute and the Flaum Eye Institute at the University of Rochester in New York, published the results of their study today (Wednesday, June 12) in the journal Science Translational Medicine.
Harnessing a benign virus for gene therapy
Three groups of researchers have successfully restored some sight to more than a dozen people with a rare disease called Leber's congenital amaurosis, which leads to complete loss of vision in early adulthood. They achieved this by inserting a corrective gene into adeno-associated viruses (AAV), a common but benign respiratory virus, and injecting the viruses directly into the retina. The photoreceptor cells take up the virus and incorporate the functional gene into their chromosomes to make a critical protein that the defective gene could not, rescuing the photoreceptors and restoring sight.
Unfortunately, the technique cannot be applied to most blinding diseases because the needle often causes retinal detachment, making the situation worse. Yet the standard AAV used in eye and other types of gene therapy cannot penetrate into tissue to reach the photoreceptors and other cells, such as retinal pigment epithelium, that need to be fixed. The retina is about 100,000 times thicker than AAV, which is about 20 nanometers across.
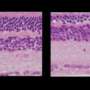
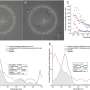
Years ago, Schaffer set out to find a way to "evolve" AAV to penetrate tissues, including eye and liver, as a way to deliver genes to specific cells. To date he has generated 100 million variants of AAV – each carrying slightly different proteins on its coat – from which he and his colleagues selected five that were effective in penetrating the retina. They then used the best of these (7m8) to transport genes to cure two types of hereditary blindness for which there are mouse models: X-linked retinoschisis, which strikes only boys and makes their retinas look like Swiss cheese; and Leber's congenital amaurosis. In each case, when injected into the vitreous humor, the AAV delivered the corrective gene to all areas of the retina and restored retinal cells nearly to normal.
When injected into the eye of a normal monkey, the virus penetrated cells spottily across the retina but almost completely in the very important fine-vision area called the fovea. Current viruses do not penetrate foveal cells at all.
Schaffer predicts that the virus can be used not only to insert genes that restore function to non-working genes, but can knock out genes or halt processes that are actively killing retina cells, which may be the case in age-related macular degeneration.
"When I first got here 14 years ago, I really had the idea or the goal that I wanted to work on problems that would have direct impact on human health, and we are now getting there," Schaffer said.
More information: "In Vivo–Directed Evolution of a New Adeno-Associated Virus for Therapeutic Outer Retinal Gene Delivery from the Vitreous," by D. Dalkara et al. Science Translational Medicine, 2013.