How mice teach us about disease
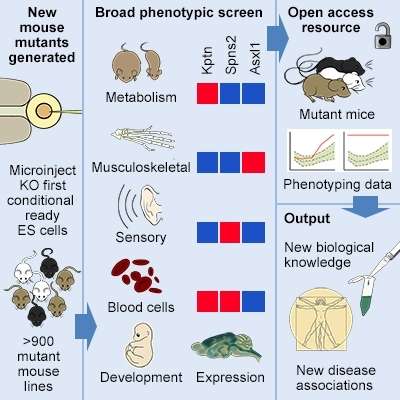
Researchers have created a large new resource of more than 900 genes switched off one-at-a-time in mice to discover which genes are important for a wide range of biological functions such as fertility or hearing.
This resource, known as the Mouse Genetics Project, screens for characteristics and early signs of disease, revealing many new functions for well-known genes, as well as for genes with no previously-known role in disease. Many of these variations in body function are likely to underlie human diseases.
The human genome has more than 20,000 identified genes, but our understanding of what they do and how disease results from their malfunction is quite limited. Studies using mice are central to understanding how genes function and how variation to genes causes disease. Mice share the vast majority of their genes with humans and researchers can use mice that have a specific gene switched off to start to unravel human disease.
The Mouse Genetics Project provides researchers and clinicians with a wealth of freely available clinical and biological information that will help find new treatment strategies and options for a wide range of diseases. Furthermore, research groups worldwide can use these new mouse lines to continue the work of investigating the fundamental mechanisms of disease, as the mouse lines are openly available to researchers.
"Our project has revealed many completely unexpected associations between genes and traits like body weight, emphasising how difficult it is to predict the importance of a gene in disease until there is a model like the mouse to give us clues," says Professor Karen Steel, lead author from King's College London and Honorary Faculty at the Wellcome Trust Sanger Institute. "There are many paths to developing a disease that aren't immediately obvious. This resource provides an efficient way to uncover these paths.
"This resource is the largest collection of mouse lines available to researchers where all other genes are virtually identical except the gene switched off. Together with the careful optimisation of their environment and the systematic analysis of a wide range of characteristics in each line, we are able to detect features that otherwise would be missed."
So far the team have individually switched off more than 900 genes in mice to understand how they function and how they relate to disease. Rather than tunnelling in on one or two potential effects, the team are systematically studying a large number of potential outcomes to build an entire catalogue of gene functions and effects. The new report published today describes the detailed analysis of the first 250 lines to undergo this systematic health screen.
The researchers discovered that newly identified and unknown genes are just as likely to underlie disease features as known genes. This finding emphasises the value of a broad approach to studying diseases rather than the current tendency of researchers to focus on well-known genes. The availability of mouse models and publicly-available information about their characteristics should encourage a broader understanding of the full extent of gene function and how genetic changes can result in disease.
The team revealed that a gene, Kptn, previously thought to be linked to deafness could actually be associated with obesity. They found that when the gene is switched off in mice fed with a high fat diet, the mice gain weight faster than those that have working copies of the gene. This is one of the many examples of genes identified by the team to have a surprising new role in disease.
Among the genes reported in detail are 26 brand new mouse models for human diseases, allowing detailed investigation of the underlying biology of these diseases. For example, variations to the gene SMS can cause a rare, inherited condition associated with both mental and physical difficulties, called Snyder-Robinson syndrome. The team switched off the same gene in mice and the results reproduced those seen in the human disease. They also detected male infertility, suggesting a new feature of the disease that may not have been recognised in humans with SMS mutations.
"Not only is the biological information openly available to the wider scientific and clinical community, but so are our mouse models," says Dr Jacqui White, first author from the Wellcome Trust Sanger Institute. "Already 447 research teams in 25 different countries across the world are using our mouse lines and taking this research to new levels. Our hope is that other research teams will take our research forward to better understand disease and develop new and effective therapies against these diseases."
"This resource is revealing a wealth of information about human disease that has great potential for improved diagnosis and treatment options," says Professor Philip Beales Professor of Medical Genetics at the Institute of Child Health, University College London, and a consultant clinical geneticist at Guy's Hospital and Great Ormond Street Hospital. "This is just the start of a long journey, but I'm excited by the research that this resource will allow us to do."
More information: DOI: 10.1016/j.cell.2013.06.022

















