October 23, 2013 report
Dendritic spines, memories, and memories of dendritic spines
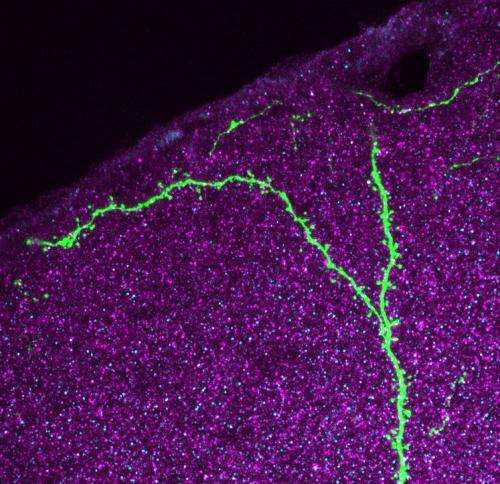
(Medical Xpress)—Nothing raises the hackles on the neck of a neurobiologist like talk of dendritic spines on neurons. These little outcroppings of membrane and contractile tissue adorn the long apical tendrils of excitatory pyramidal cells in the cortex. They are found on neurons in subcortical organs as well, but for now anyway, the mission is to first define their function where they can best be seen. Researchers at the Institute for Molecular Pathology in Vienna have just published a paper in PNAS where they report their recent findings about pyramidal cell spines in live, behaving mice. They seek to define the role of spines, and plasticity in general in the role of learning and memory.
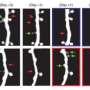
The tool which lets neurobiologists see spines is the two-photon microscope. Several labs have harnessed this device previously to try to determine how much change, or shall we say movement, occurs in spines during memory formation. Other studies have focused on motor areas of the cortex, and looked at spines during the acquisition of motor skills. The Vienna researchers were interested in looking at the currently hip topic of how the recall of a memory, either just after learning it (the consolidation epoch), or at some time much later, affects the memory itself, and its hypothesized molecular substrate. If only that were something even remotely feasible.
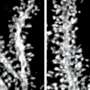
Fortunately, the researchers were not deterred and actually succeeded in watching the brains of mice over extended periods of time during which experiments designed to probe auditory-cued fear conditioning were performed. Needless to say, spines came and went. There really is no perfect metric to define a spine. Generally the concept of "I know one when I see one" suffices. An important question about spines is how long on average do they persist, relative to the transitional period where they are either coming to be, or are being disassembled. This is a difficult thing to quantify because there is no clear guidance on how we should assign a number to a particular configuration of 2d membrane surface contorted through a given 3d space.
Even if such a number was on hand, there is a further complication—spines are not just passively immotile electrical compartments, but rather, they move, just like everything else in the brain—and probably even a fair bit faster. Spine twitching has been known to be a thing for a long time, even Francis Crick had a fair bit to say about that particular curiosity several decades ago. If spines, and the larger neuropil topology as a whole are actually being re-scaled to the tune of a 60% cerebral volume change over any given day—as we now would seem to be assured—is there any convenient candle in the structure that can be used to define it?
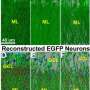
Embryologists often watch the nuclei of cells to get a better picture of what is going on inside developing tissue. This is particularly useful for those synticial tissues, like muscle or bone precursors, where cell borders have been dissolved. In neurons though, there are far more features of interest than those that could be characterized just by a nuclei. A full spine load on a pyramidal cell might push upwards of several thousand spines. While not every spine contains them, there is one structure that is the most predictive of the current state of a spine, and where it is headed—the presence of its resident mitochondria. The saying, "know a neuron by its mitochondria," may not have caught on entirely yet, but understanding spines, via mitochodrial proxy may be the best option we have now going forward.
If memory is be to gauged by the mass appearance or loss of spine, there is one caveat. Anyway you slice it, the cortical grey matter is already full. If you add spines, you either need to take away something else, or thicken the cortex and collapse the ventricles and subdural space. If instead you try to hand pick which spines correspond to which skills and memories, what you are essentially doing, is trying to estimate the movements of termites by the behavior of planetary bodies.
Similarly, there is one point that may merit highlighting as far as general methodology of memory, that may or may not be fair. If both crude intimations in c-fos expression, and high-tech two-photon probing of synaptic structures as fine as a spine are offered alongside each other as comparable molecular level indicators of memory, there may be some cause for concern. In taking our 16 year-old out for a driving lesson, we do not simultaneously discuss both how to walk, and how to pilot a passenger plane in the same breath.
More information: Dynamics of dendritic spines in the mouse auditory cortex during memory formation and memory recall, PNAS, Published online before print October 22, 2013, DOI: 10.1073/pnas.1312508110
Abstract
Long-lasting changes in synaptic connections induced by relevant experiences are believed to represent the physical correlate of memories. Here, we combined chronic in vivo two-photon imaging of dendritic spines with auditory-cued classical conditioning to test if the formation of a fear memory is associated with structural changes of synapses in the mouse auditory cortex. We find that paired conditioning and unpaired conditioning induce a transient increase in spine formation or spine elimination, respectively. A fraction of spines formed during paired conditioning persists and leaves a long-lasting trace in the network. Memory recall triggered by the reexposure of mice to the sound cue did not lead to changes in spine dynamics. Our findings provide a synaptic mechanism for plasticity in sound responses of auditory cortex neurons induced by auditory-cued fear conditioning; they also show that retrieval of an auditory fear memory does not lead to a recapitulation of structural plasticity in the auditory cortex as observed during initial memory consolidation.
© 2013 Medical Xpress