November 19, 2013 report
Imaging the magnetically stimulated brain
(Medical Xpress)—MRI scanners have steadily increased in power, giving researchers ever finer-grained snapshots of the brain in action. However just as modern day fighters can pull high G turns that would drain consciousness from a human pilot, the maximum field strength that humans, (and the implants now inside many of them) might be subjected to, is similarly bounded. As these limits approach, the psycho-imagers that wield these impressive tools have begun to consolidate their key findings into a mission statement solid enough to print on enterprise plaques. In order to bolster their claims, there has been a push to confirm hypothesized functional networks in the brain, by stimulating brain activity directly and comparing the results to the so-called resting state (rsMRI) activity. Neurobiologist Karl Deisseroth, and his mostly Stanford colleges, have now used transcranial magnetic stimulation of the brain (TMS) to probe these networks on subjects as they lie inside the bore of an MRI magnet. Their results, just published in PNAS, suggest that when combined with precise stimulation protocols, important details about cognition and memory might continue to be extracted amidst an impending plateau in the basic physics of MRI imaging.
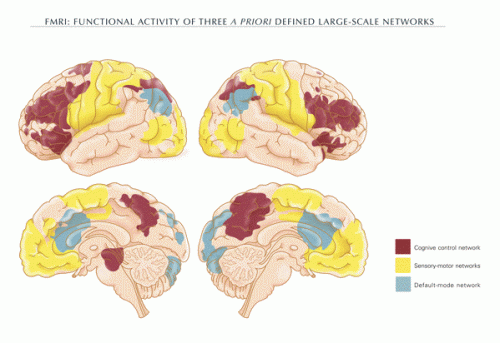
The main findings of this study hinge of the existence of three functional networks in brain that have been previously defined by the community. They are the fronto-parietal central executive network (CEN), the cingulo-opercular salience network (SN), and the medial prefrontal-medial parietal default mode networks (DMN). While many of us may be none the wiser for having known them, there are still plenty of good things to be learned in pursuit of their elusive character.
Towards the end of better defining these networks, Deisseroth had previously combined optogenetics with MRI in a technique known as OfMRI. With this powerful method he could selectively stimulate a class of CaMKIIa-expressing excitatory neurons, either in the neocortex or thalamus, of rats suitable modified by virus. For humans however, as in the present study, the researchers used the somewhat less specific magnetic stimulation protocol. The basic premise here is that depending on how and where it is applied, TMS can either excite or inhibit the neurons or fibers within its field of influence. Therein lies the rub. While now approved for an ever-expanding list of therapeutic opportunities, it is difficult to establish the definitive, and uniform, excitation or inhibition within any given localized area.

By exciting or inhibiting different network nodes, the researchers were able to show that the DMN is under inhibitory control of specific nodes within the CN. As the authors note, inferring causality from neuroimaging data requires that standard correlative methods be combined with direct manipulation of activity—and that they seem to have done. Although the field of TMS has advanced considerably, and its practicitioners now enjoy amenities like precise robotic positioners, and advanced software like the Brainsight II package, the ability to precisely toggle a "node" might be called in to question when what constitutes the node itself is vague.
In summary, the recent blossoming of new MRI techniques, not only those listed above, but many others including diffusion dtMRI, functional connectivity fcMRI, or event-related efMRI, in a sense parallels what is going on now in the expanding field of experimental RNA technology. Most of us have been familiar with the standard triplet family of RNA - messenger, transfer, and ribosomal RNAs. Now, the appreciation and availability of a Cambrian explosion of new interfering and activating RNA tools like small hairpin shRNA, small nuclear snRNA, and non-coding or pRNA, has created a situation where the complex and overlapping nomenclature, at least to the non-specialist looking in, threatens to collapse under its own weight. In moving forward, a more rigorous definition of MRI protocols and analysis techniques may help install greater confidence into studies involving things like default mode networks of the brain.
More information: Causal interactions between fronto-parietal central executive and default-mode networks in humans, PNAS, Published online before print November 18, 2013, DOI: 10.1073/pnas.1311772110
Abstract
Information processing during human cognitive and emotional operations is thought to involve the dynamic interplay of several large-scale neural networks, including the fronto-parietal central executive network (CEN), cingulo-opercular salience network (SN), and the medial prefrontal-medial parietal default mode networks (DMN). It has been theorized that there is a causal neural mechanism by which the CEN/SN negatively regulate the DMN. Support for this idea has come from correlational neuroimaging studies; however, direct evidence for this neural mechanism is lacking. Here we undertook a direct test of this mechanism by combining transcranial magnetic stimulation (TMS) with functional MRI to causally excite or inhibit TMS-accessible prefrontal nodes within the CEN or SN and determine consequent effects on the DMN. Single-pulse excitatory stimulations delivered to only the CEN node induced negative DMN connectivity with the CEN and SN, consistent with the CEN/SN's hypothesized negative regulation of the DMN. Conversely, low-frequency inhibitory repetitive TMS to the CEN node resulted in a shift of DMN signal from its normally low-frequency range to a higher frequency, suggesting disinhibition of DMN activity. Moreover, the CEN node exhibited this causal regulatory relationship primarily with the medial prefrontal portion of the DMN. These findings significantly advance our understanding of the causal mechanisms by which major brain networks normally coordinate information processing. Given that poorly regulated information processing is a hallmark of most neuropsychiatric disorders, these findings provide a foundation for ways to study network dysregulation and develop brain stimulation treatments for these disorders.
© 2013 Medical Xpress